A Levels Chemistry (9701)•9701/13/M/J/18
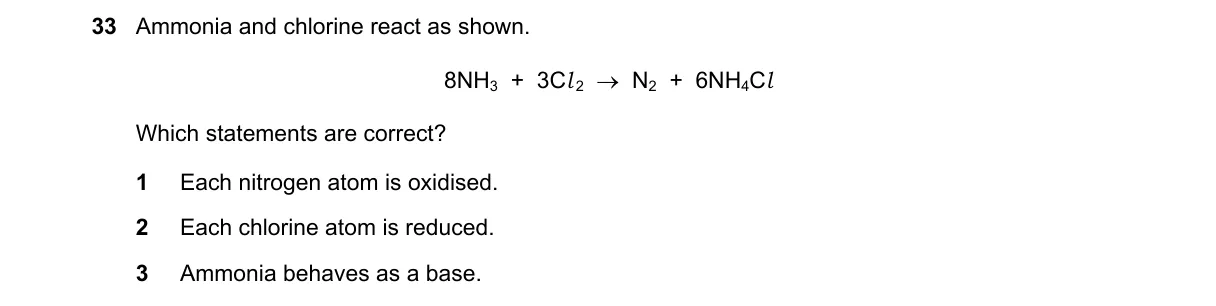
Explanation
Reaction Analysis: Redox States and Chemical Behavior
Steps:
- Calculate oxidation state of N: -3 in NH3, 0 in N2 (increase indicates oxidation).
- Calculate oxidation state of Cl: 0 in Cl2, -1 in HCl (decrease indicates reduction).
- Examine reaction products: Formation of HCl suggests acid-base interaction where NH3 acts as proton acceptor.
- Evaluate statements against definitions of reduction, oxidation, and base behavior.
Why C is correct:
- Ammonia (NH3) behaves as a base by accepting H+ ions, consistent with Lewis/Brønsted-Lowry definition, leading to HCl formation.
Why the others are wrong:
- A: Nitrogen is oxidized (state from -3 to 0), not reduced.
- B: Chlorine is reduced (state from 0 to -1), not oxidized.
- D: Not enough information (choice incomplete).
Final answer: C
Topic: Nitrogen and sulfur
Practice more A Levels Chemistry (9701) questions on mMCQ.me