A Levels Chemistry (9701)•9701/13/M/J/18
Explanation
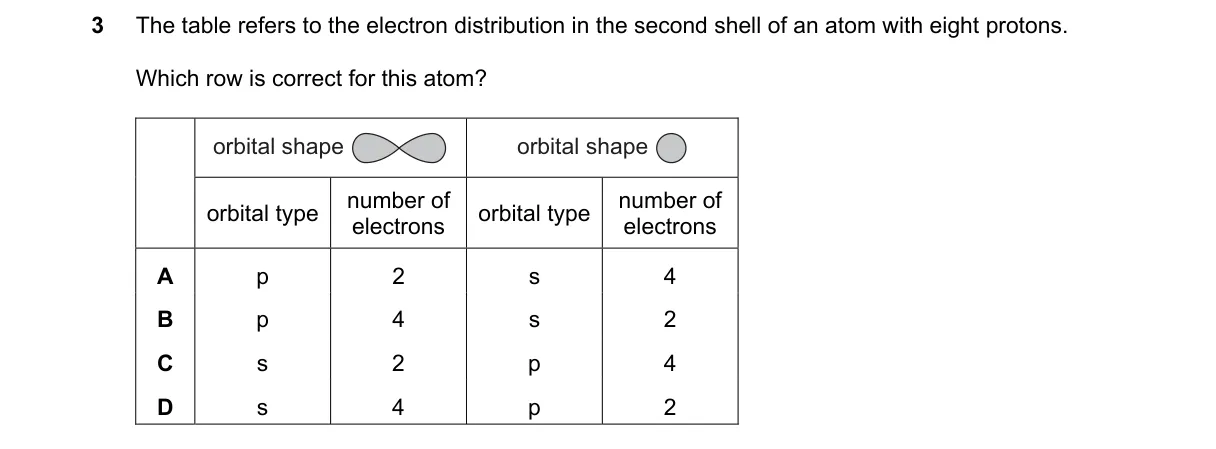
Electron configuration in the second shell of oxygen
Steps:
- Atom with 8 protons is neutral oxygen (8 electrons).
- Full configuration: 1s² 2s² 2p⁴ (follows Aufbau principle).
- Second shell (n=2) includes 2s (max 2 electrons) and 2p (max 6 electrons) subshells.
- Distribution: 2s² (s 2) and 2p⁴ (p 4), totaling 6 electrons.
Why D is correct:
- s 2 p 4 follows the order of filling (s before p) and Pauli exclusion principle, with s holding exactly 2 electrons.
Why the others are wrong:
- A: Incorrect order (p before s) and s exceeds maximum 2 electrons.
- B: s subshell cannot hold 4 electrons (maximum 2 per orbital, 2 orbitals).
- C: Incorrect order (p before s) and violates subshell capacities.
Final answer: D
Topic: Atomic structure
Practice more A Levels Chemistry (9701) questions on mMCQ.me