A Levels Chemistry (9701)•9701/12/M/J/18
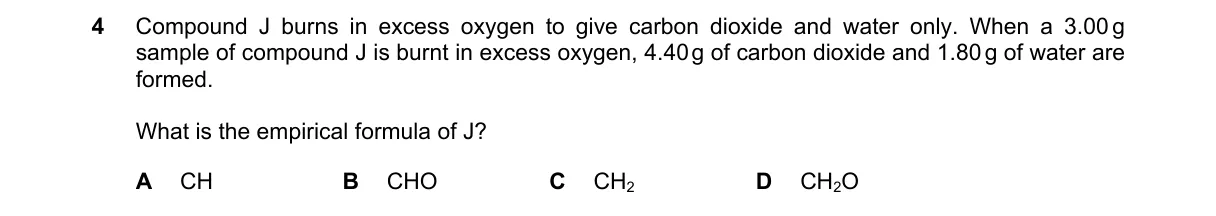
Explanation
Combustion analysis finds empirical formula from C, H, O masses in products
Steps:
- Moles C = 4.00 g CO₂ / 44 g/mol = 0.091 mol
- Moles H = (1.80 g H₂O / 18 g/mol) × 2 = 0.20 mol
- Mass O = 3.00 g - (0.091 mol × 12 g/mol) - (0.20 mol × 1 g/mol) = 1.71 g
- Moles O = 1.71 g / 16 g/mol = 0.107 mol; divide by 0.091 gives C:H:O ≈ 1:2:1, so CH₂O
Why D is correct:
- Mole ratio ≈1:2:1 defines CH₂O as the simplest whole-number empirical formula from % composition.
Why the others are wrong:
- A. CH: H:C ratio 1:1, but data gives ~2:1
- B. CHO: H:C ratio 1:1, but data gives ~2:1
- C. CH₂: No oxygen, but 1.71 g O found in J
Final answer: D
Topic: Atoms, molecules and stoichiometry
Practice more A Levels Chemistry (9701) questions on mMCQ.me