A Levels Chemistry (9701)•9701/11/M/J/18
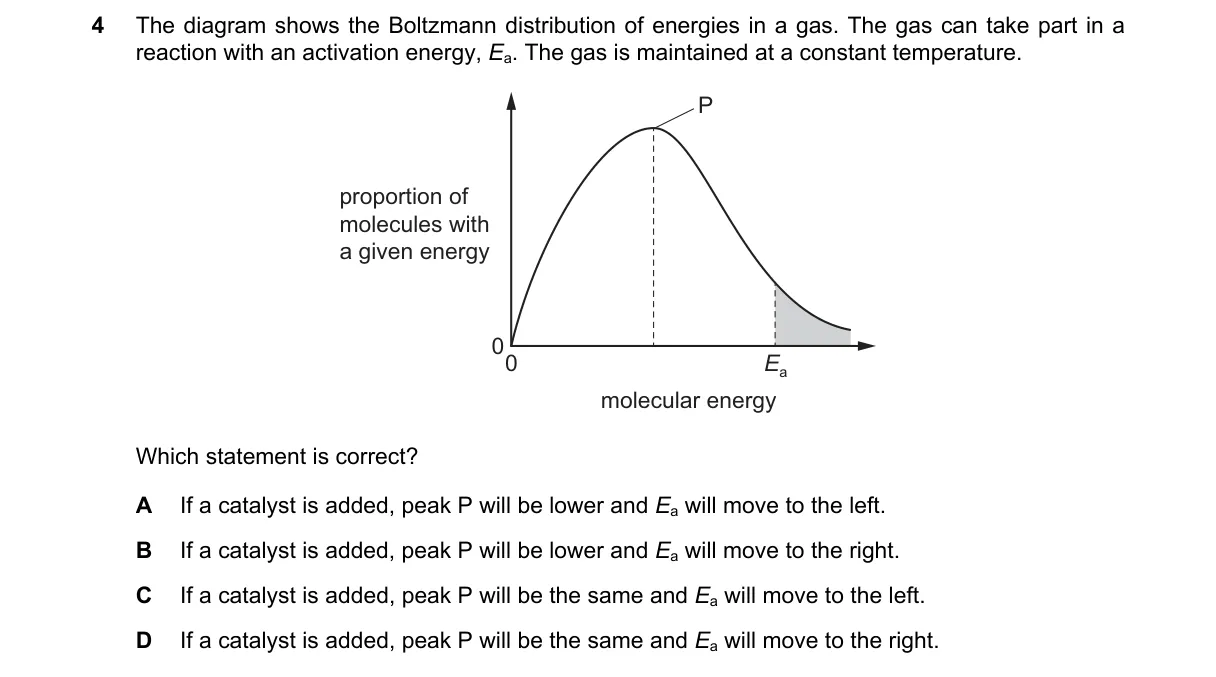
Explanation
Catalyst lowers activation energy without altering energy distribution
Steps:
- Boltzmann distribution plots molecular energies at fixed temperature; peak P is at most probable energy.
- Constant temperature means distribution shape and peak height remain unchanged by catalyst.
- Activation energy E is marked as a vertical line on the energy axis to the right of peak P.
- Catalyst reduces E, shifting the line leftward to lower energy, increasing reacting molecules.
Why B is correct:
- Catalysts lower activation energy via alternative pathway (Arrhenius equation: rate ∝ e^{-Ea/RT}), moving E left while constant T keeps peak P identical.
Why the others are wrong:
- A: Catalyst does not lower peak P or increase E.
- C: E moves left, not right.
- D: Peak P unchanged, not lower.
Final answer: B
Topic: Reaction kinetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me