A Levels Biology (9700)•9700/12/M/J/21
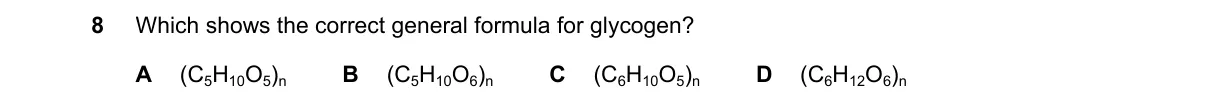
Explanation
Glycogen's Polymer Formula
Steps:
- Glycogen is a branched polysaccharide made of glucose units linked by glycosidic bonds.
- Each glucose monomer (C₆H₁₂O₆) loses H₂O during polymerization, forming a repeating unit of C₆H₁₀O₅.
- The general formula represents the polymer as (repeating unit)_n, where n is the degree of polymerization.
- Confirm the formula matches standard biochemistry references for glycogen.
Why C is correct:
- (C₆H₁₀O₅)_n is the established formula for glycogen, reflecting the dehydrated glucose units in its α-1,4 and α-1,6 linkages.
Why the others are wrong:
- A uses incorrect subscript notation, implying a different hydration state.
- B omits the polymer indicator, suggesting a monomer.
- D includes an extra oxygen, altering the empirical ratio.
Final answer: C
Topic: Carbohydrates and lipids
Practice more A Levels Biology (9700) questions on mMCQ.me