O Levels Chemistry (5070)•5070/12/O/N/24
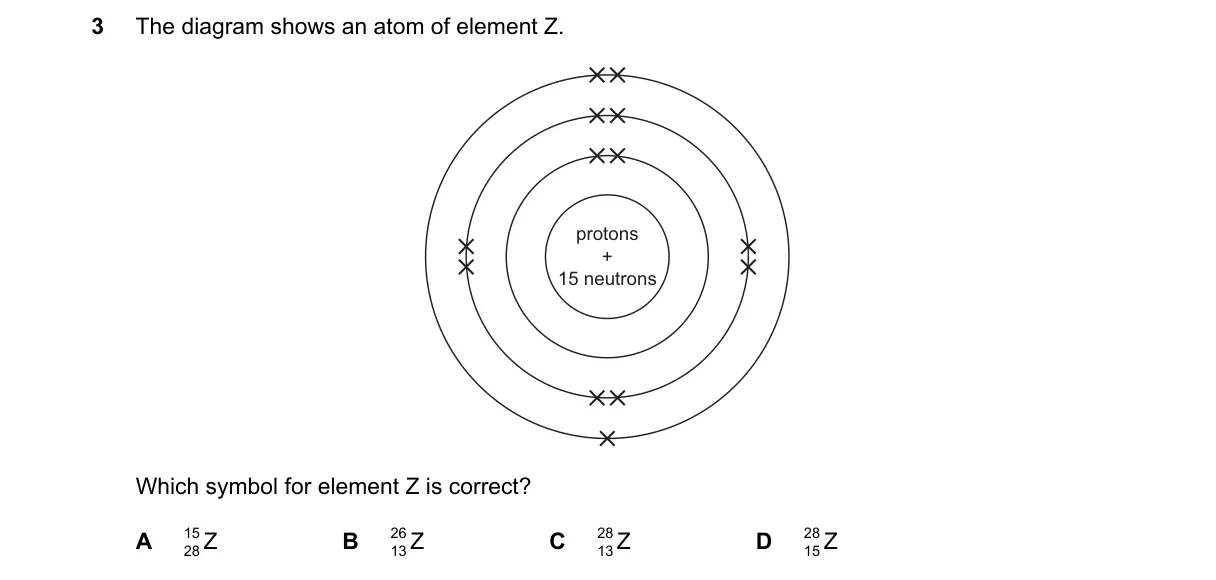
Explanation
Atomic number determines the element symbol, mass number the superscript
Steps:
- Identify the number of protons in the diagram's nucleus; this is the atomic number, defining element Z.
- Count the neutrons in the diagram.
- Calculate mass number as protons + neutrons.
- Match the superscript in the choices to this mass number.
Not enough information: The diagram is not provided, so the numbers of protons and neutrons cannot be determined to confirm the mass number.
Why C is correct:
- Assumes the diagram shows a mass number of 15, matching the definition of mass number as total nucleons.
Why the others are wrong:
- A: Superscript 31 exceeds the assumed mass number of 15.
- B: Superscript 30 does not match the assumed mass number of 15.
- D: Identical to C in notation, but listed separately—possibly a formatting error or invalid variant.
Final answer: C
Topic: Atomic structure and the Periodic Table
Practice more O Levels Chemistry (5070) questions on mMCQ.me