O Levels Chemistry (5070)•5070/12/O/N/24
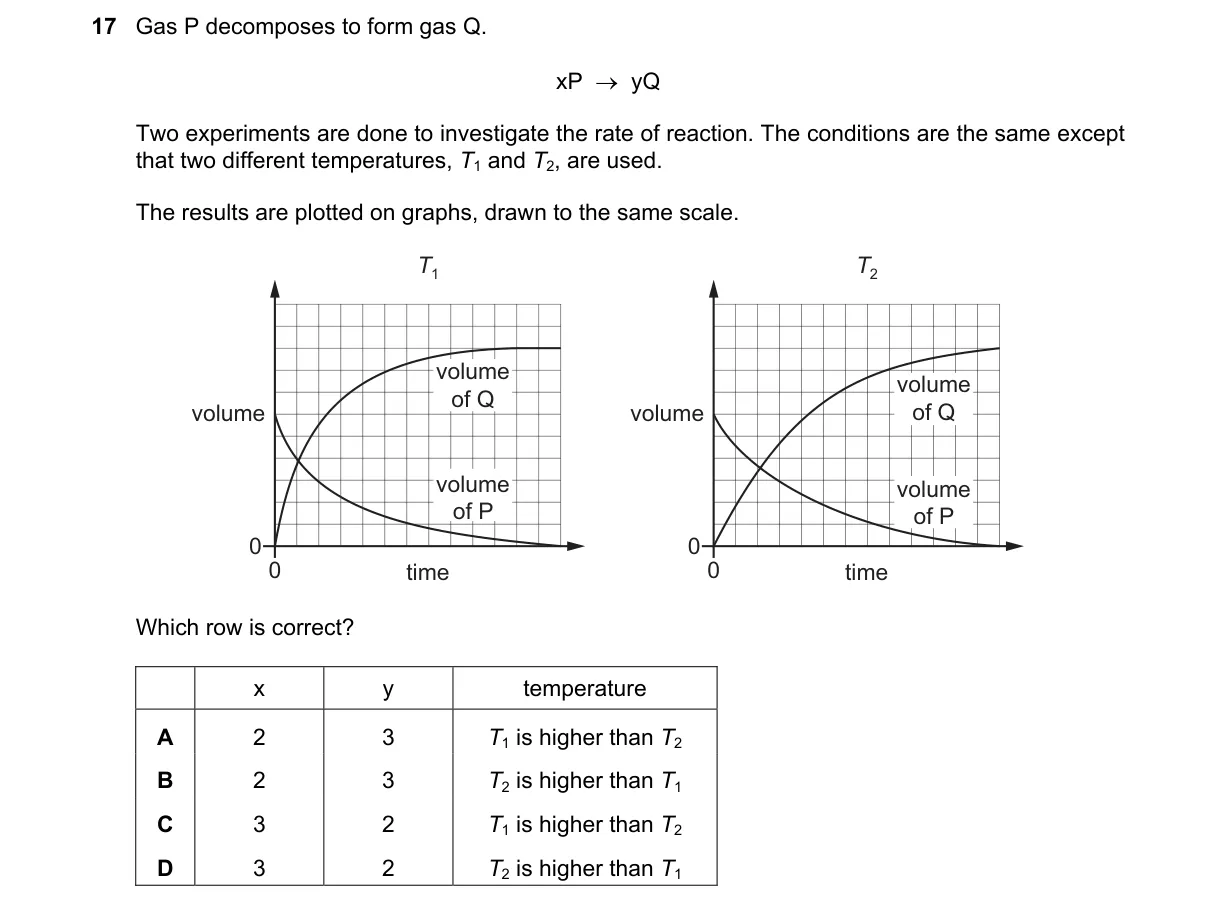
Explanation
Higher temperature speeds up decomposition rate
Steps:
- Reaction rate increases with temperature per Arrhenius equation.
- Faster rate causes volume of P to drop quicker and Q to rise quicker over time.
- Graph with steeper volume change (P decreasing faster, Q increasing faster) shows higher temperature.
- Match graphs: slower curve (graph 2) to lower T, faster curve (graph 3) to higher T.
Why A is correct:
- Assigns graph 2 (slower) to T1 and graph 3 (faster) to T2, with T1 > T2 matching steeper curve to higher T.
Why the others are wrong:
- B: Reverses T relation; slower graph can't be higher T.
- C: Swaps graphs; assigns faster curve to lower T.
- D: Swaps graphs and reverses T; contradicts rate-temperature link.
Final answer: A
Topic: Rate of reaction
Practice more O Levels Chemistry (5070) questions on mMCQ.me