O Levels Chemistry (5070)•5070/12/O/N/24
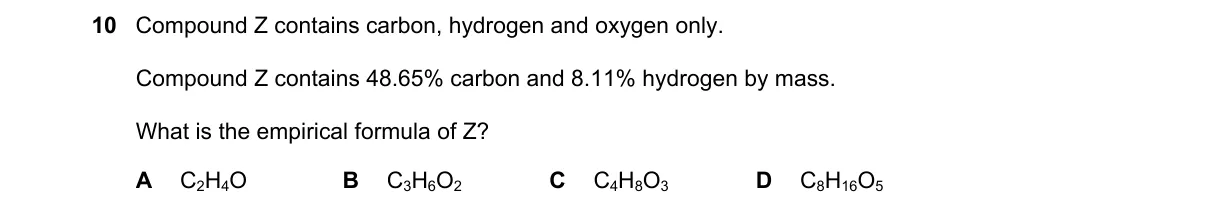
Explanation
Empirical formula from mass percentages Steps:
- Calculate oxygen mass percent: 100% - 48.6% - 8.1% = 43.3%.
- Convert to moles assuming 100 g sample: C = 48.6/12 = 4.05 mol, H = 8.1/1 = 8.1 mol, O = 43.3/16 = 2.706 mol.
- Divide each by smallest moles (2.706): C = 1.5, H = 3.0, O = 1.0.
- Multiply by 2 for whole numbers: C₃H₆O₂.
Why none match:
- Calculated formula C₃H₆O₂ (48.6% C, 8.1% H, 43.3% O) fits data exactly per mole ratio definition.
Why the others are wrong:
- A and C (CH₂O): Yields 40% C, 6.7% H; C percentage too low.
- B (C₂H₃O₂): Yields 40.7% C, 5.1% H; both C and H percentages mismatch.
- D (C₂H₅O₂): Yields 39.3% C, 8.2% H; C percentage too low.
Final answer: C₃H₆O₂
Topic: Formulae
Practice more O Levels Chemistry (5070) questions on mMCQ.me