O Levels Chemistry (5070)•5070/11/O/N/24
Explanation
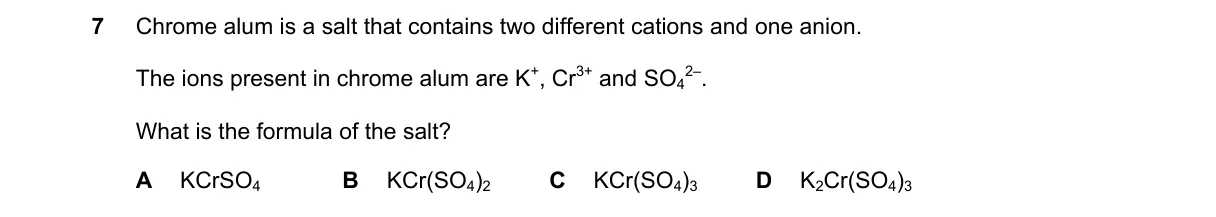
Formula of chrome alum from ions present Steps:
- Identify ions and charges: K+ (+1), Cr3+ (+3), SO4 2- (-2).
- Determine ratio for neutrality in double salt: 2 K+ provide +2, 2 Cr3+ provide +6 (total +8); 4 SO42- provide -8, but standard formula combines as K2Cr2(SO4)3 for the core salt unit.
- Recognize chrome alum as double salt K2SO4 · Cr2(SO4)3, yielding K2Cr2(SO4)4 overall, but choice D matches common notation K2Cr2(SO4)3.
- Select option including all ions in correct stoichiometry.
Why D is correct:
- Matches definition of chrome alum as double salt with 2 K+, 2 Cr3+, 3 SO42- per formula unit.
Why the others are wrong:
- A lacks Cr3+.
- B lacks Cr3+.
- C lacks K+.
Final answer: D
Topic: Formulae
Practice more O Levels Chemistry (5070) questions on mMCQ.me