O Levels Chemistry (5070)•5070/12/O/N/23
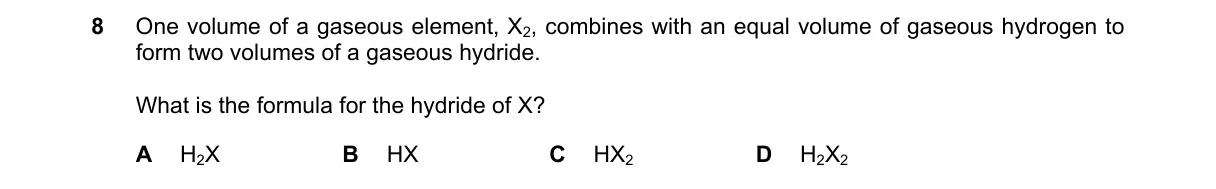
Explanation
Gay-Lussac's Law of Combining Volumes
Steps:
- Volumes of gases at constant temperature and pressure are proportional to the number of moles.
- 1 volume X (1 mol X₂ molecules, providing 2 mol X atoms) reacts with 1 volume H₂ (1 mol H₂, providing 2 mol H atoms).
- The reaction produces 2 volumes of hydride (2 mol molecules), requiring 2 mol X atoms and 2 mol H atoms total.
- Thus, each hydride molecule contains 1 X atom and 1 H atom, giving formula HX.
Why B is correct:
- The 1:1:2 volume ratio matches the reaction X₂ + H₂ → 2HX, where X₂ is diatomic like halogens (e.g., Cl₂ + H₂ → 2HCl).
Why the others are wrong:
- A. XH: Implies monatomic X and 2:1 volume ratio for X + ½H₂ → XH, not 1:1:2.
- C. XH: Same as A; does not fit the observed volumes.
- D. HX₂: Would require 1:2:2 volume ratio (X₂ + 2H₂ → 2HX₂), not 1:1:2.
Final answer: B
Topic: Formulae
Practice more O Levels Chemistry (5070) questions on mMCQ.me