O Levels Chemistry (5070)•5070/12/O/N/22
Explanation
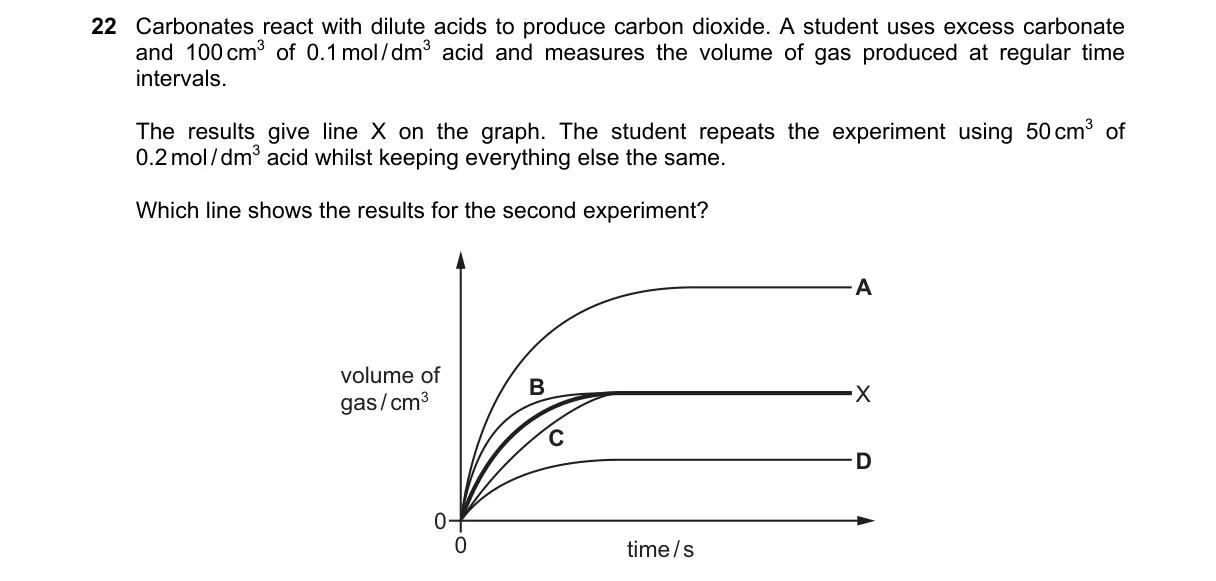
Same acid moles yield equal total CO2, but higher concentration speeds up the reaction rate
Steps:
- Calculate acid moles in first experiment: 100 cm³ (0.1 dm³) × 0.1 mol/dm³ = 0.01 mol.
- Second experiment: 50 cm³ (0.05 dm³) × 0.2 mol/dm³ = 0.01 mol, identical amount.
- Same carbonate amount means equal total CO2 produced in both.
- Higher acid concentration in second experiment increases reaction rate, producing steeper volume-time curve.
Why B is correct:
- B shows steeper initial slope to same final volume, as rate law states reaction rate ∝ acid concentration.
Why the others are wrong:
- A: Shows half total volume, ignoring equal moles of acid.
- C: Depicts slower rate, contradicting higher concentration effect.
- D: Indicates different final volume, violating equal reactant moles.
Final answer: B
Topic: Rate of reaction
Practice more O Levels Chemistry (5070) questions on mMCQ.me