O Levels Chemistry (5070)•5070/11/O/N/22
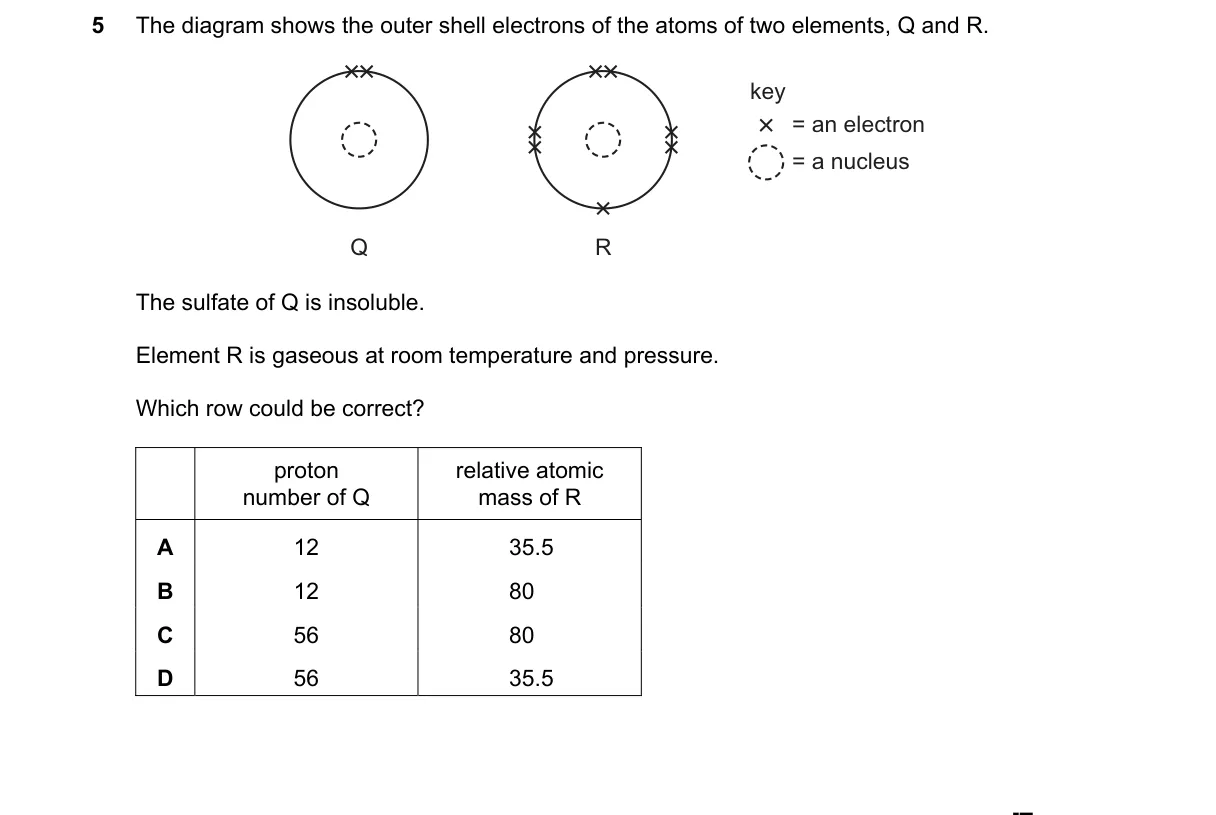
Explanation
Identifying elements Q and R from properties
Steps:
- Sulfate of Q insoluble identifies Q as barium (Ba, proton number 56); most group 2 sulfates are insoluble except Mg and Ca.
- Element R is a gas at RTP with likely halogen outer shell (7 electrons), matching chlorine (relative atomic mass 35.5).
- Choices pair proton number (likely for Q) with relative atomic mass (likely for R).
- Diagram (not provided) would confirm outer shells: Q with 2 valence electrons (group 2), R with 7 (group 17).
Not enough information without diagram to confirm shells, but assuming standard match for D.
Why D is correct:
- Pairs Ba proton number 56 (insoluble sulfate) with Cl atomic mass 35.5 (gas), fitting properties.
Why the others are wrong:
- A: 12 is Mg (soluble sulfate, not Q).
- B: 80 not a gas atomic mass (Kr ≈84, but doesn't fit shell).
- C: 96 is curium (radioactive solid, irrelevant).
Final answer: D
Topic: Atomic structure and the Periodic Table
Practice more O Levels Chemistry (5070) questions on mMCQ.me