O Levels Chemistry (5070)•5070/11/O/N/22
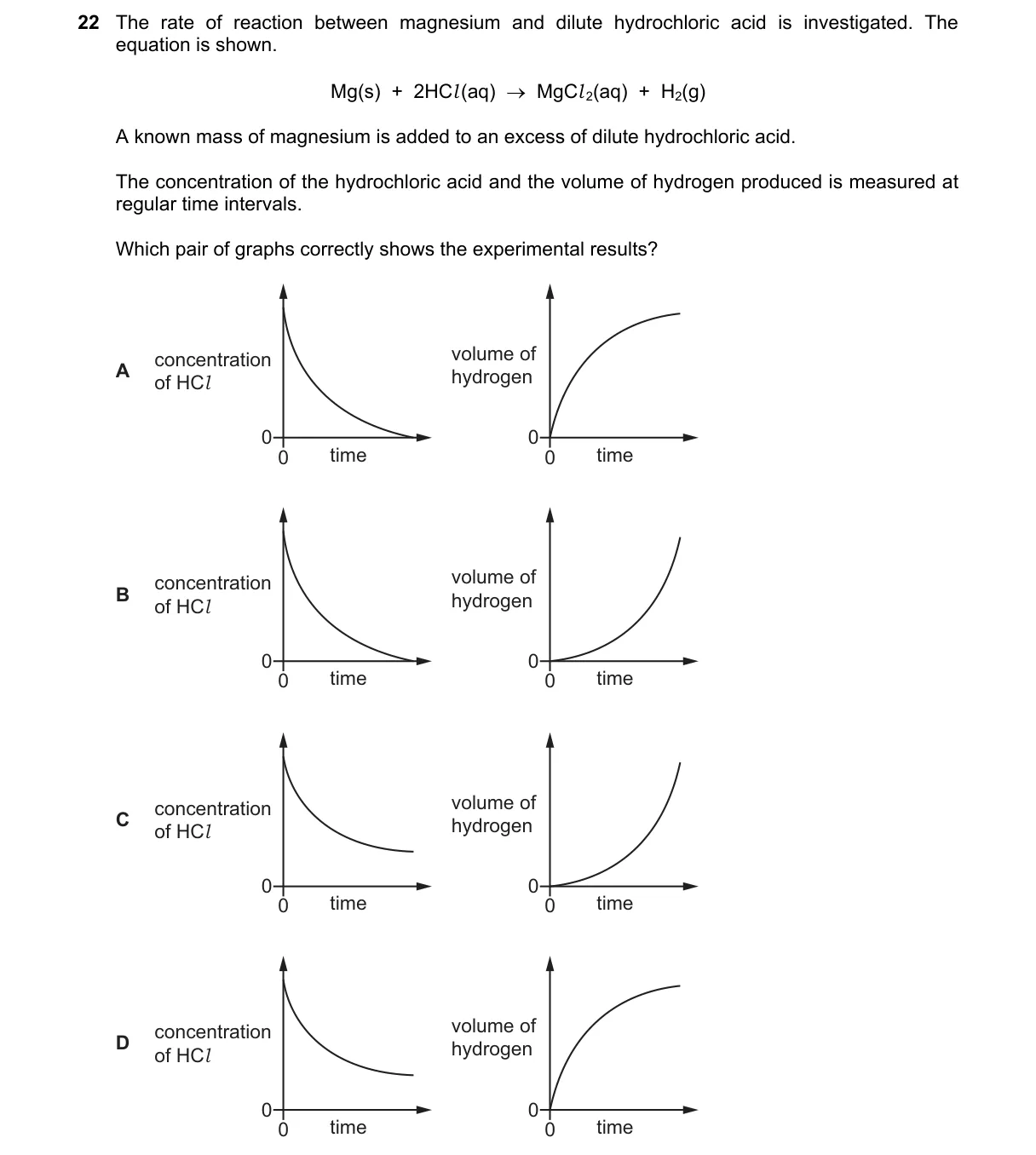
Explanation
Reaction monitoring via product volume and reactant concentration
Steps:
- Reaction consumes HCl, so its concentration decreases over time from initial value toward zero (or excess if present).
- Reaction produces H2 gas, so its volume increases from zero toward a maximum stoichiometric limit.
- Measurements at regular intervals yield curves: [HCl] declines nonlinearly; H2 volume rises nonlinearly to plateau.
- Correct graphs pair a decreasing [HCl]-time curve with an increasing H2 volume-time curve.
Why D is correct:
- Displays [HCl] decreasing and H2 volume increasing asymptotically, matching Le Chatelier's principle and integrated rate laws for reactant depletion and product accumulation.
Why the others are wrong:
- A: Shows linear changes, ignoring slowing rate as reactants deplete.
- B: Inverts curves (H2 decreasing, [HCl] increasing), contradicting reaction direction.
- C: Both curves plateau immediately, implying no reaction progress.
Final answer: D
Topic: Rate of reaction
Practice more O Levels Chemistry (5070) questions on mMCQ.me