O Levels Chemistry (5070)•5070/12/O/N/20
Explanation
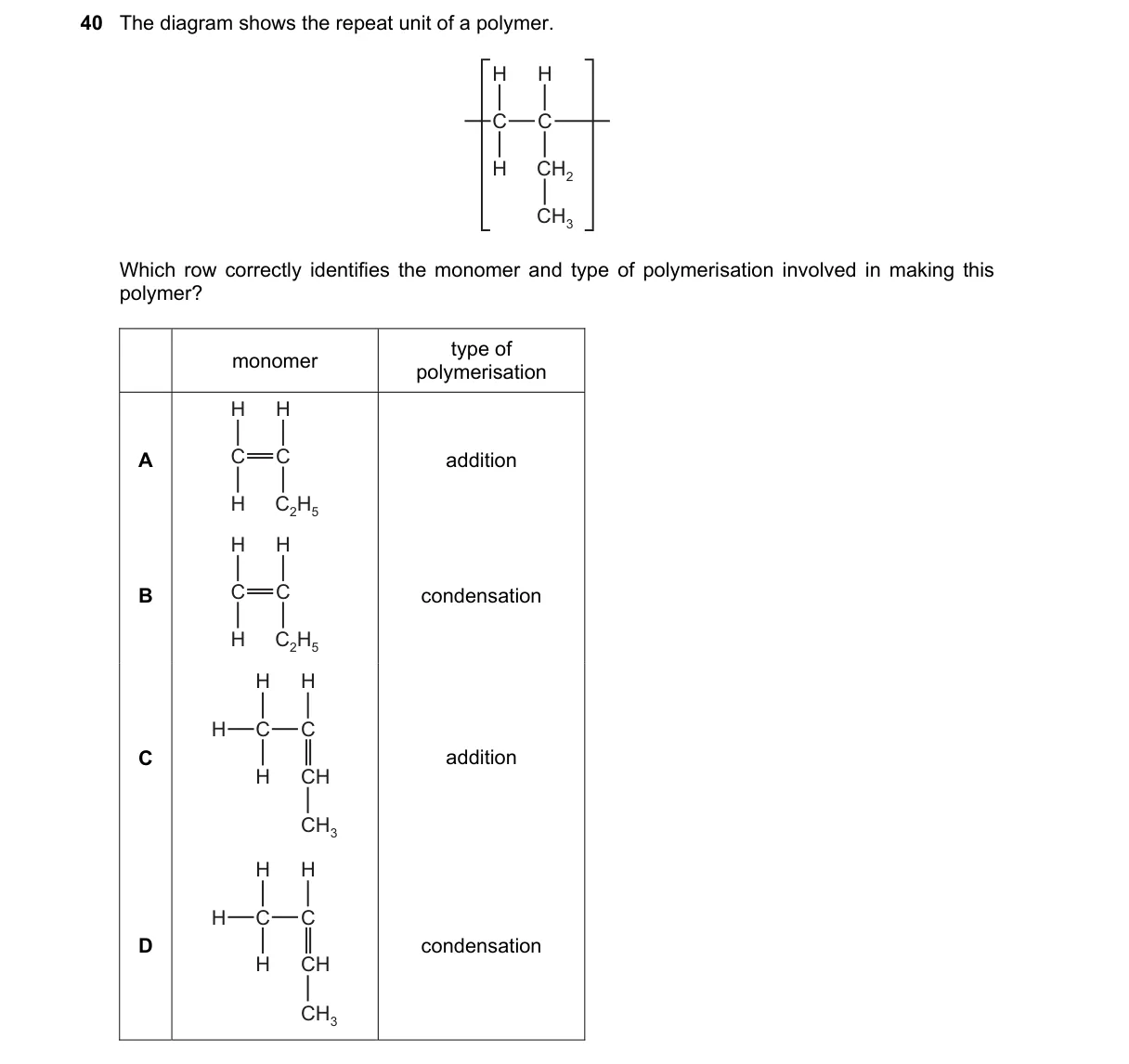
Matching repeat unit to monomer and polymerization type
Steps:
- Examine the diagram's repeat unit: -[CH2-CH2]-.
- Recognize this as the structure from ethylene monomer.
- Confirm addition polymerization: double bond opens without losing atoms.
- Rule out alternatives by structure mismatch or wrong mechanism.
Why A is correct:
- The repeat unit -[CH2-CH2]- matches addition polymerization of H2C=CH2, where the alkene adds across the double bond per the addition reaction definition.
Why the others are wrong:
- B: Condensation polymerization eliminates small molecules like water; this polymer shows no such loss.
- C: H3C-CH=CH2 (propene) yields -[CH2-CH(CH3)]- repeat unit, not -[CH2-CH2]-.
- D: H3C-CH3 (ethane) lacks a reactive double bond for polymerization; condensation requires functional groups like -OH or -COOH.
Final answer: A
Topic: Polymers
Practice more O Levels Chemistry (5070) questions on mMCQ.me