O Levels Chemistry (5070)•5070/11/O/N/20
Explanation
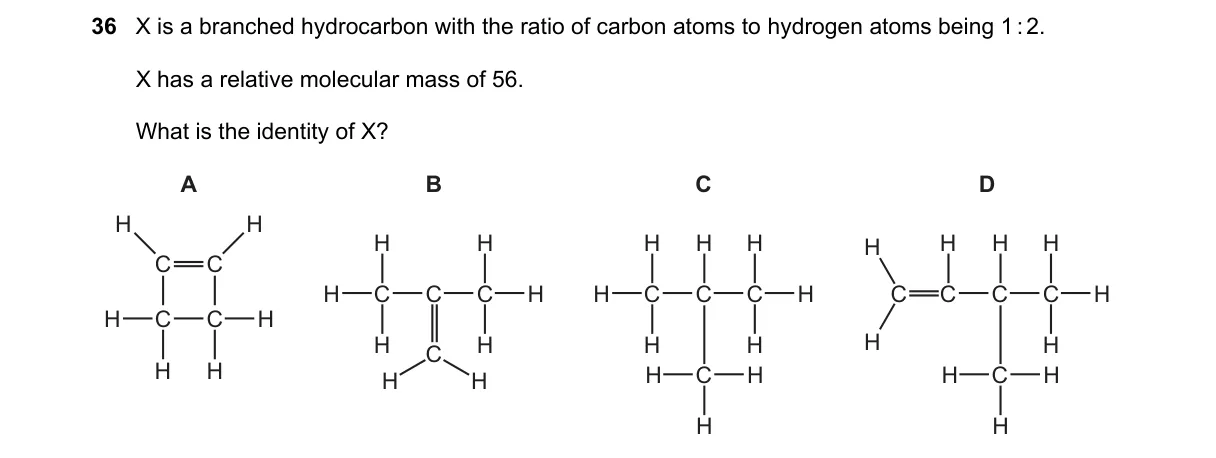
Molecular formula from C:H ratio and mass determines branched alkene structure
Steps:
- C:H ratio of 1:2 matches alkene general formula C_nH_{2n}.
- Relative molecular mass of 56 gives 12n + 2n = 14n = 56, so n=4 and formula is C_4H_8.
- C_4H_8 isomers include straight-chain and branched alkenes; branched specifies 2-methylpropene.
- Option B depicts 2-methylpropene, fitting all criteria.
Why B is correct:
- 2-Methylpropene is the branched C_4H_8 alkene with formula mass (124 + 18 = 56) matching the given ratio per alkene rule C_nH_{2n}.
Why the others are wrong:
- A: Straight-chain but-1-ene, lacks branching.
- C: Cyclobutane, cyclic not branched.
- D: Butane (C_4H_{10}), wrong ratio (1:2.5) and mass (58).
Final answer: B
Topic: Alkenes
Practice more O Levels Chemistry (5070) questions on mMCQ.me