O Levels Chemistry (5070)•5070/11/O/N/20
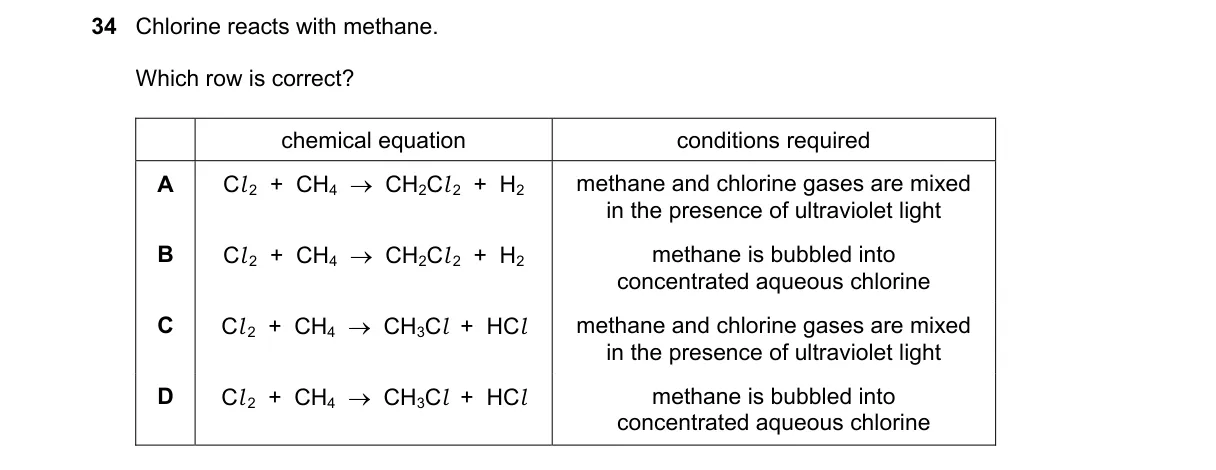
Explanation
Free radical substitution of methane
Steps:
- Identify the balanced equation: Cl₂ + CH₄ → CH₃Cl + HCl.
- Confirm products: substitution yields CH₃Cl and HCl, not H₂.
- Recall conditions: requires gaseous reactants and UV light for initiation.
- Match option with correct equation and conditions.
Why C is correct:
- Matches the free radical mechanism where UV light generates Cl• radicals, leading to CH₃Cl + HCl as per the substitution reaction formula.
Why the others are wrong:
- A: Produces H₂ instead of HCl, violating the substitution product.
- B: Produces H₂ instead of HCl and uses aqueous conditions, which do not support free radical initiation.
- D: Uses aqueous chlorine, which prevents the gas-phase UV-driven reaction.
Final answer: C
Topic: Alkanes
Practice more O Levels Chemistry (5070) questions on mMCQ.me