O Levels Chemistry (5070)•5070/11/O/N/20
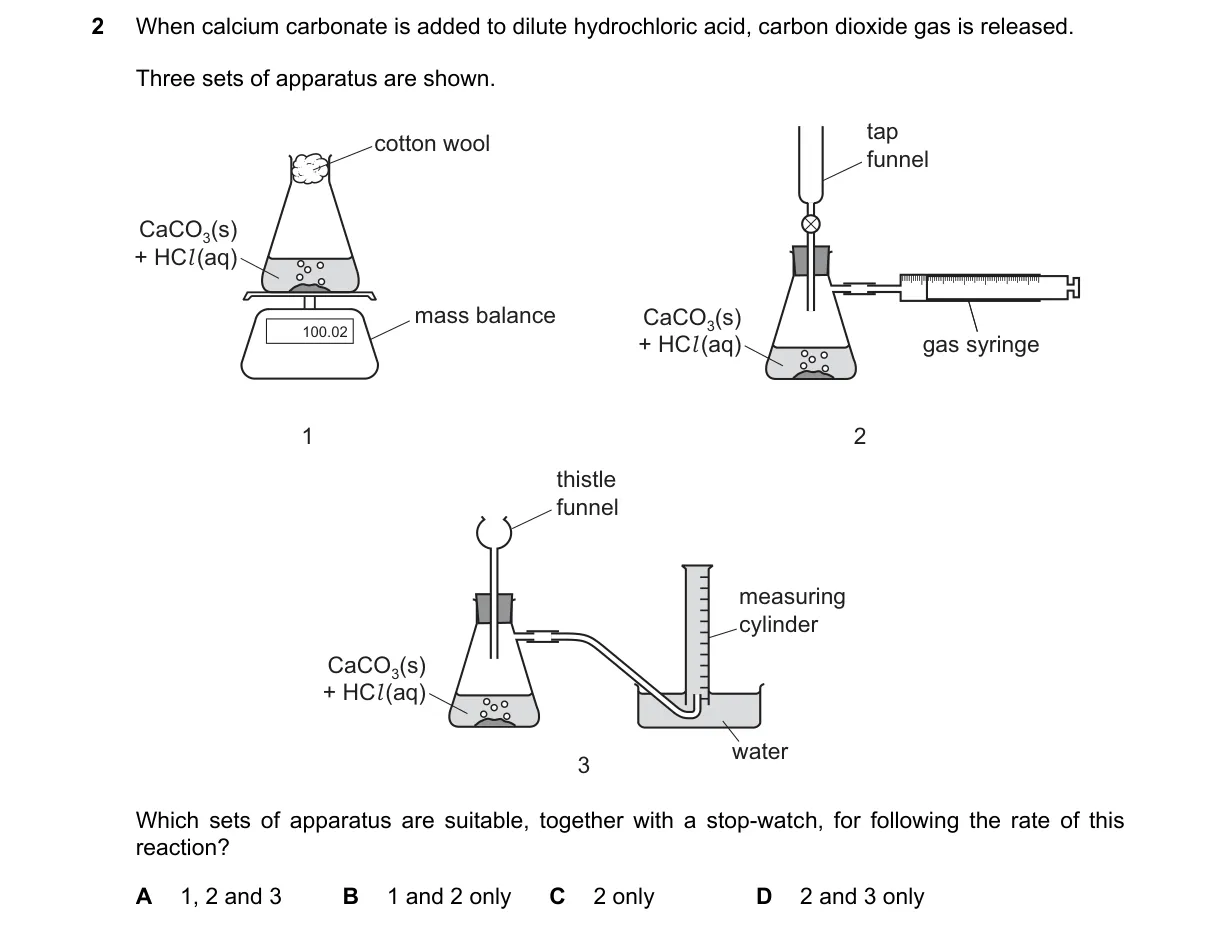
Explanation
Measuring Reaction Rate via Gas Production or Mass Loss
Steps:
- Reaction CaCO3 + 2HCl → CaCl2 + H2O + CO2 produces CO2 gas; rate measured by gas volume or mass loss over time with stopwatch.
- Set 1 uses gas syringe to collect and measure CO2 volume directly against time.
- Set 2 uses balance to track mass decrease from CO2 escape over time.
- Set 3 lacks direct gas volume or mass measurement, unsuitable for rate tracking.
Why B is correct:
- Sets 1 and 2 align with rate law definition: rate ∝ Δ[product]/Δt, where gas volume or mass loss quantifies CO2 production proportionally.
Why the others are wrong:
- A includes unsuitable set 3, which cannot track gas evolution.
- C omits set 1, missing a valid gas volume method.
- D includes unsuitable set 3 and omits set 1, lacking full valid options.
Final answer: B
Topic: Rate of reaction
Practice more O Levels Chemistry (5070) questions on mMCQ.me