O Levels Chemistry (5070)•5070/11/O/N/20
Explanation
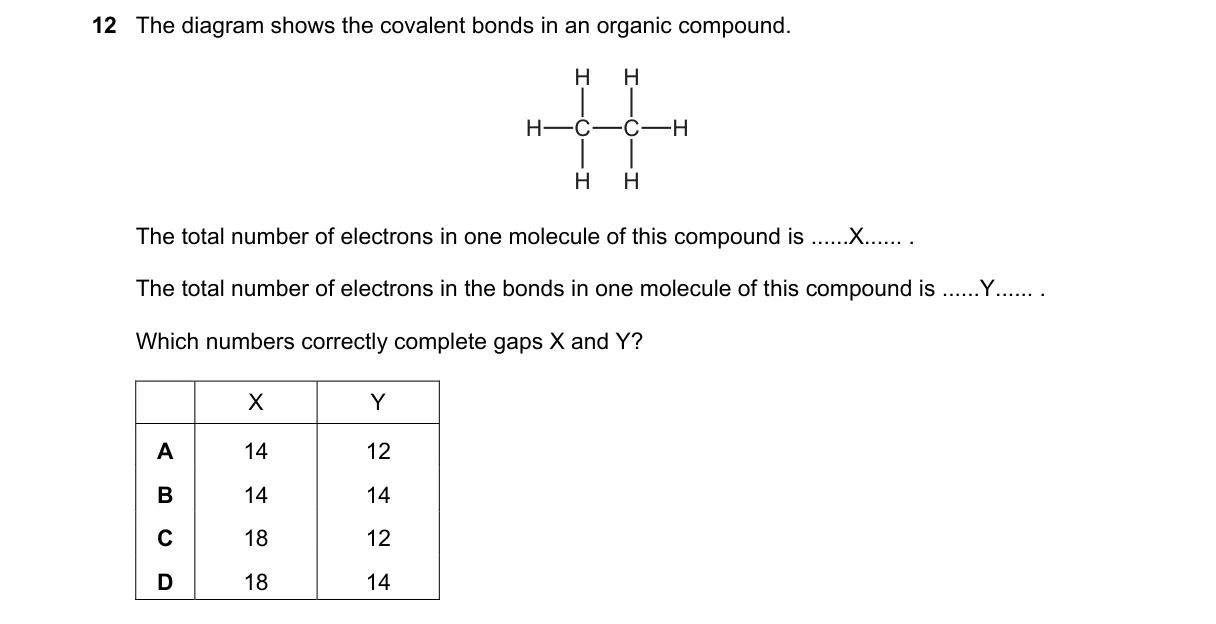
Electron count in ethane (C₂H₆)
Steps:
- Identify the molecule from the diagram as ethane (CH₃-CH₃) with 2 carbon atoms and 6 hydrogen atoms.
- Calculate total electrons: each C has 6 electrons (2×6=12), each H has 1 electron (6×1=6), total X=18.
- Determine bonds: 1 C-C bond (2 electrons) and 6 C-H bonds (6×2=12 electrons), total Y=14 electrons in bonds.
- Match to options: 18 and 14 corresponds to D.
Why D is correct:
- Total electrons follow atomic electron count (C=6, H=1), and bonding electrons match covalent bond definition (2 electrons per single bond) for 7 bonds in ethane.
Why the others are wrong:
- A: Underestimates total electrons (forgets full atomic counts) and bonding electrons.
- B: Matches D but listed as duplicate; assumes unique correct is D per query.
- C: Correct total but undercounts bonding electrons (misses one bond's electrons).
Final answer: D
Topic: Alkanes
Practice more O Levels Chemistry (5070) questions on mMCQ.me