O Levels Chemistry (5070)•5070/12/O/N/19
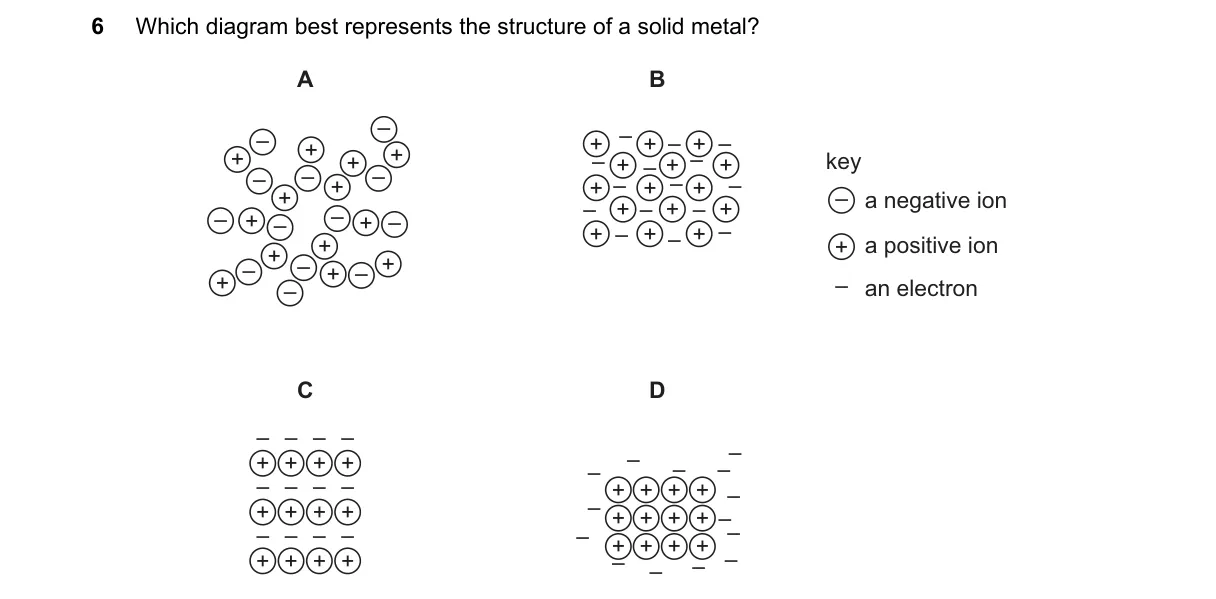
Explanation
Metallic lattice structure in solid metals
Steps:
- Recall that solid metals consist of a regular arrangement of positive metal ions.
- Identify the key feature: delocalized electrons surround the ions, forming a "sea" of electrons.
- Compare diagrams: look for one showing cations in a lattice with free-moving electrons, not bonds or molecules.
- Select the diagram matching this metallic bonding model.
Why B is correct:
- It depicts positive ions in a fixed lattice surrounded by delocalized electrons, aligning with the metallic bond definition where electrons are free to move.
Why the others are wrong:
- A shows localized electron pairs, representing covalent bonding.
- C illustrates alternating positive and negative ions, typical of ionic crystals.
- D displays discrete molecules, characteristic of molecular solids.
Final answer: B
Topic: Metallic bonding
Practice more O Levels Chemistry (5070) questions on mMCQ.me