O Levels Chemistry (5070)•5070/12/O/N/19
Explanation
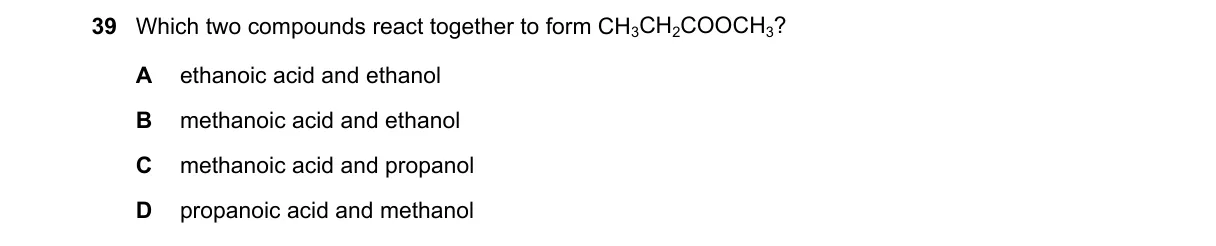
Esterification Reaction Forms Methyl Ethanoate Steps:
- Identify the target compound: CH3COOCH3 is methyl ethanoate (methyl acetate), an ester.
- Recall esterification: Carboxylic acid + alcohol → ester + water, where the ester formula combines the acid's acyl group (CH3CO-) and alcohol's alkyl group (-CH3).
- Match the acid: CH3CO- comes from ethanoic acid (CH3COOH).
- Match the alcohol: -CH3 comes from methanol (CH3OH).
Why none are correct:
- No option pairs ethanoic acid with methanol; all produce different esters.
Why the others are wrong:
- A: Forms ethyl ethanoate (CH3COOC2H5).
- B: Forms ethyl methanoate (HCOOC2H5).
- C: Forms propyl methanoate (HCOOC3H7).
- D: Forms methyl propanoate (CH3CH2COOCH3).
Not enough information: Options do not include the correct pair.
Final answer: None
Topic: Carboxylic acids
Practice more O Levels Chemistry (5070) questions on mMCQ.me