O Levels Chemistry (5070)•5070/12/O/N/19
Explanation
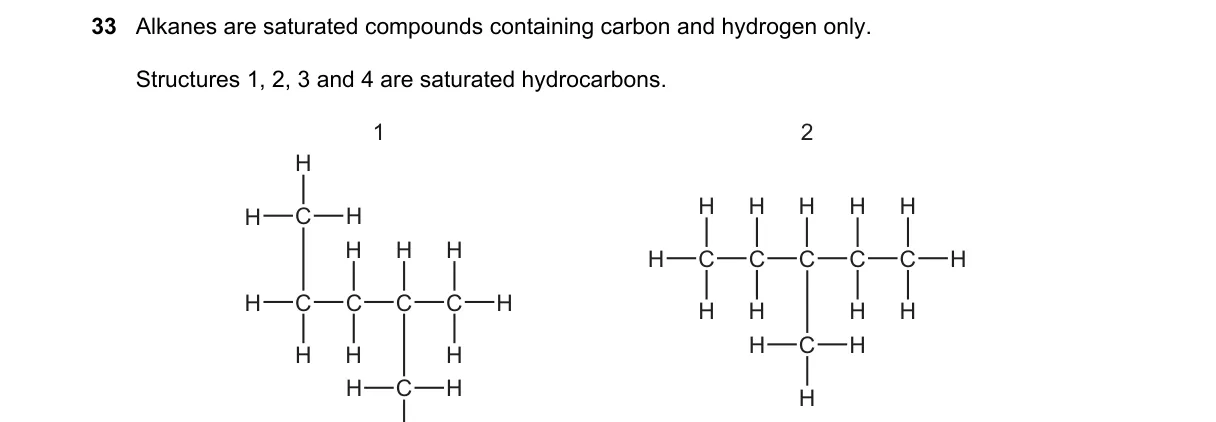
Saturated hydrocarbons have only single C-C bonds
Steps:
- Identify saturated hydrocarbons as those with carbon and hydrogen atoms connected solely by single bonds, no double or triple bonds.
- Examine structure 1: all C-C bonds are single → saturated.
- Examine structure 2: all C-C bonds are single → saturated.
- Examine structure 3: contains a C=C double bond → unsaturated.
- Examine structure 4: all C-C bonds are single → saturated.
Why A is correct:
- Option A correctly identifies structures 1, 2, and 4 as having only single C-C bonds, matching the definition of saturated hydrocarbons (general formula CₙH₂ₙ₊₂ for alkanes).
Why the others are wrong:
- B includes structure 3, which has a double bond and is unsaturated.
- C excludes structure 1, which is saturated, and includes unsaturated structure 3.
- D excludes structure 2, which is saturated, and includes unsaturated structure 3.
Final answer: A
Topic: Alkanes
Practice more O Levels Chemistry (5070) questions on mMCQ.me