O Levels Chemistry (5070)•5070/11/O/N/19
Explanation
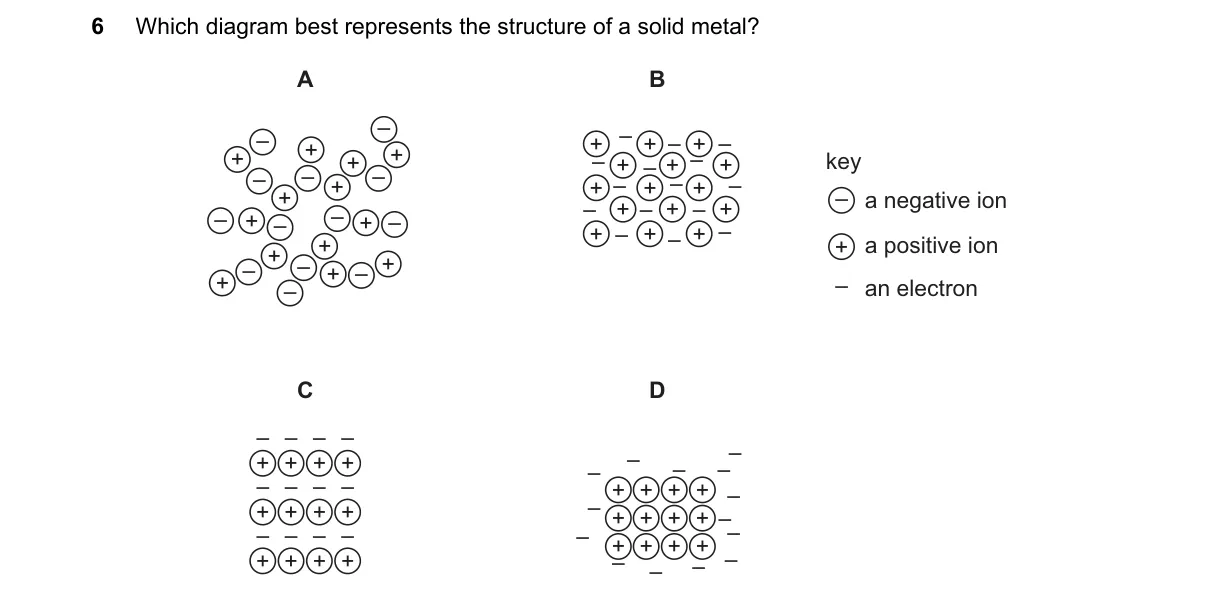
Metallic Bonding in Solid Metals Steps:
- Identify solid metal structure as a giant lattice of positive ions in a regular arrangement.
- Recognize delocalized valence electrons forming a "sea" that binds the ions.
- Eliminate diagrams showing discrete bonds or molecules, as metals lack them.
- Select the diagram illustrating packed ions with surrounding electron cloud.
Why B is correct:
- It shows a lattice of positive ions immersed in delocalized electrons, matching the metallic bonding model where electrons are free to move.
Why the others are wrong:
- A: Depicts discrete molecular structures, typical of covalent compounds, not metals.
- C: Illustrates alternating positive and negative ions, characteristic of ionic solids like salts.
- D: Shows loosely packed atoms without electron delocalization, resembling a gas or simple molecular solid.
Final answer: B
Topic: Metallic bonding
Practice more O Levels Chemistry (5070) questions on mMCQ.me