O Levels Chemistry (5070)•5070/11/O/N/19
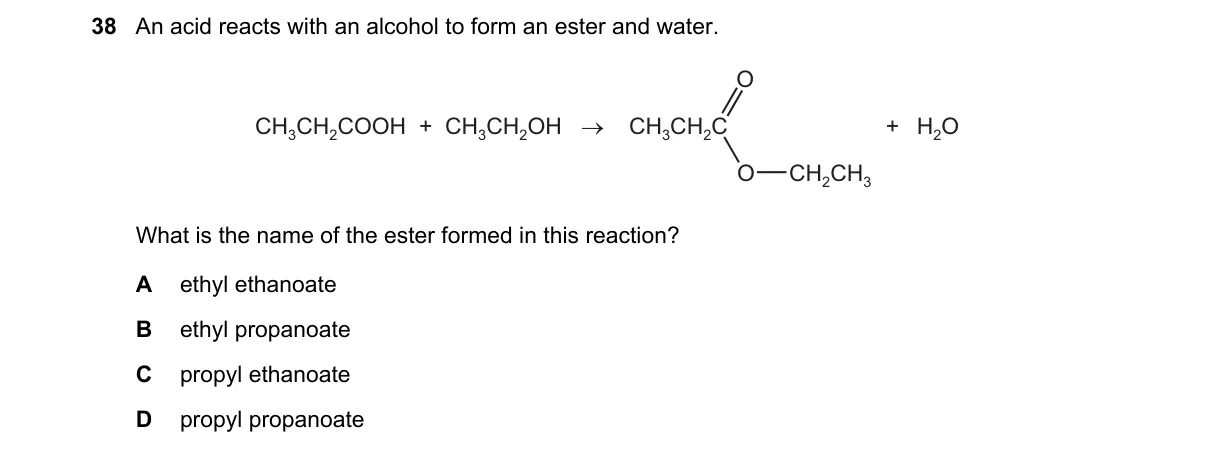
Explanation
Ester naming convention Steps:
- Identify the carboxylic acid: CH3COOH is ethanoic acid, contributing the "ethanoate" part.
- Identify the alcohol: CH3CH2OH is ethanol, contributing the "ethyl" alkyl group.
- Combine as alkyl alkanoate: ethyl from alcohol + ethanoate from acid.
- Verify formula: CH3COOCH2CH3 matches ethyl ethanoate structure.
Why A is correct:
- Follows IUPAC rule: ester name is alkyl (from alcohol) + alkanoate (from acid's carboxylate).
Why the others are wrong:
- B: "Propanoate" implies propanoic acid (C2H5COOH), but acid is ethanoic (CH3COOH).
- C: "Propyl" implies propanol (C3H7OH), but alcohol is ethanol (C2H5OH).
- D: Combines incorrect "propyl" and "propanoate" from wrong acid and alcohol.
Final answer: A
Topic: Naming organic compounds
Practice more O Levels Chemistry (5070) questions on mMCQ.me