O Levels Chemistry (5070)•5070/11/O/N/19
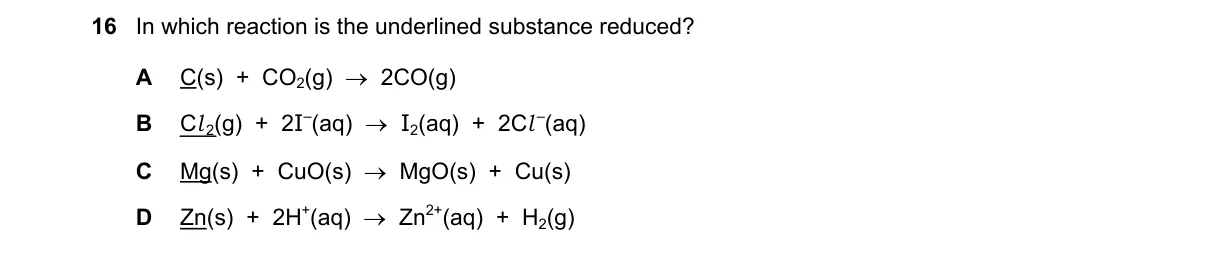
Explanation
Reduction involves decrease in oxidation number
Steps:
- Recall reduction: gain of electrons or drop in oxidation state of the underlined reactant.
- For each option, assign oxidation numbers to the underlined substance before/after reaction.
- Check if oxidation number decreases (reduced) or increases (oxidized).
- Identify the option where underlined substance's oxidation number decreases.
Why B is correct:
- In Mg(s) + CuO(s) → MgO(s) + Cu(s), underlined CuO has Cu at +2; in Cu(s), it's 0—oxidation number decreases, so reduced.
Why the others are wrong:
- A: Underlined Na(l) at 0 becomes Na+ at +1 in NaCl—increases, oxidized.
- C: Underlined Zn(s) at 0 becomes Zn²⁺ at +2—increases, oxidized.
- D: Underlined Fe(s) at 0 becomes Fe²⁺ at +2—increases, oxidized.
Final answer: B
Topic: Redox
Practice more O Levels Chemistry (5070) questions on mMCQ.me