O Levels Chemistry (5070)•5070/11/O/N/19
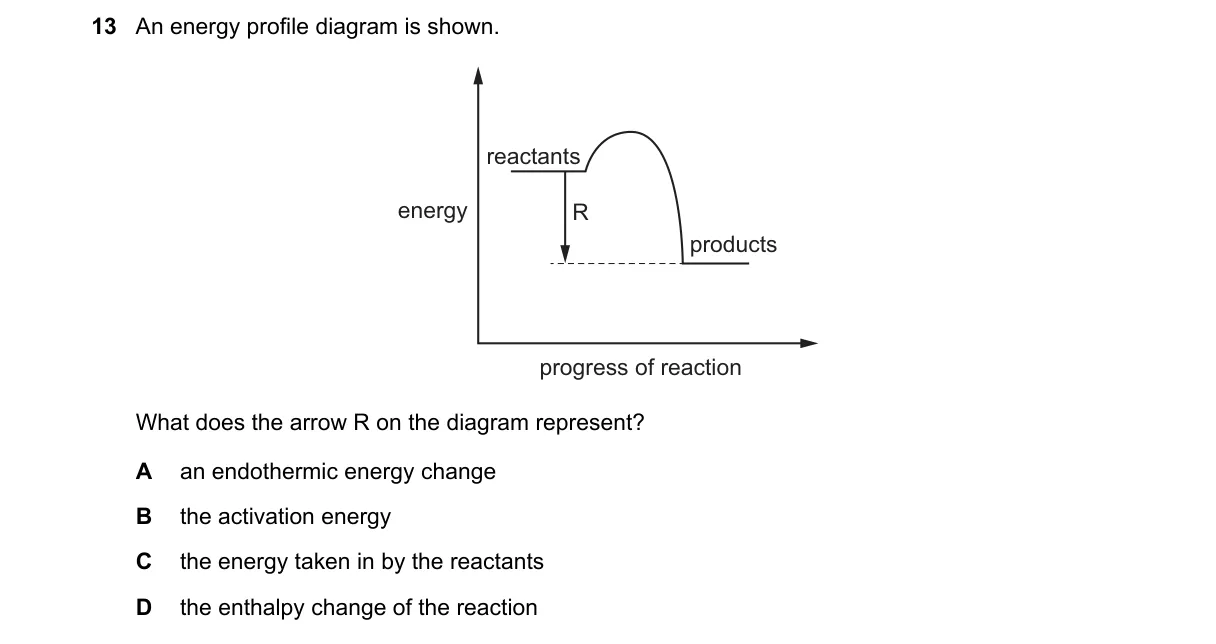
Explanation
Arrow in Energy Diagrams Shows Enthalpy Change
Steps:
- Recognize the diagram as an energy profile for a chemical reaction, plotting energy vs. reaction progress.
- Identify the arrow as the vertical difference between reactant and product energy levels.
- Recall that this difference quantifies the net energy absorbed or released during the reaction.
- Match the arrow to the definition of enthalpy change (ΔH), the heat transferred at constant pressure.
Why D is correct:
- Enthalpy change (ΔH) is defined as the difference in enthalpy between products and reactants (ΔH = H_products - H_reactants), directly represented by the arrow's length and direction in energy diagrams.
Why the others are wrong:
- A: Endothermic change indicates ΔH > 0 (upward arrow), but the arrow itself represents the magnitude and sign of ΔH, not just endothermic processes.
- B: Activation energy is the hump from reactants to the transition state, not the overall arrow between levels.
- C: Reactant energy is the starting horizontal line, not the arrow showing change.
Final answer: D
Topic: Exothermic and endothermic reactions
Practice more O Levels Chemistry (5070) questions on mMCQ.me