O Levels Chemistry (5070)•5070/12/O/N/18
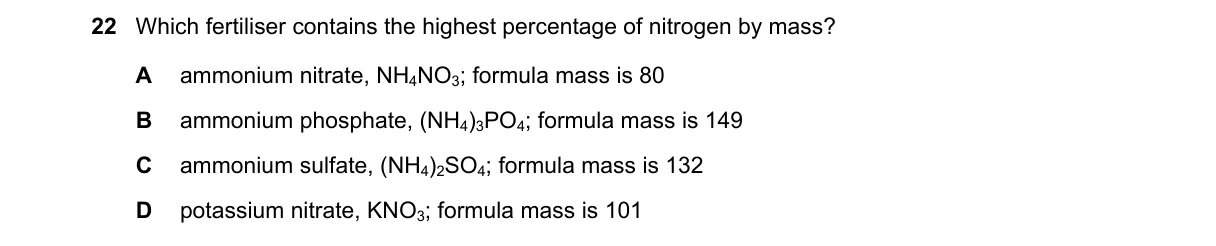
Explanation
Calculate % nitrogen by mass for each fertilizer
Steps:
- Percentage nitrogen = (total atomic mass of N atoms / formula mass) × 100.
- For A (NH₄NO₃): 2 N atoms × 14 = 28; 28/80 × 100 = 35%.
- For B ((NH₄)₃PO₄): 3 N × 14 = 42; 42/149 × 100 ≈ 28%.
- For C ((NH₄)₂SO₄): 2 N × 14 = 28; 28/132 × 100 ≈ 21%.
- For D (KNO₃): 1 N × 14 = 14; 14/101 × 100 ≈ 14%.
Why A is correct:
- Ammonium nitrate has the highest %N (35%) because it contains two N atoms in a low formula mass, maximizing N proportion per mass unit.
Why the others are wrong:
- B has lower %N (28%) due to three N atoms diluted by heavier phosphate group.
- C has lower %N (21%) from sulphate's higher mass relative to two N atoms.
- D has lowest %N (14%) with only one N atom and added potassium mass.
Final answer: A
Topic: Fertilisers
Practice more O Levels Chemistry (5070) questions on mMCQ.me