O Levels Chemistry (5070)•5070/12/M/J/23
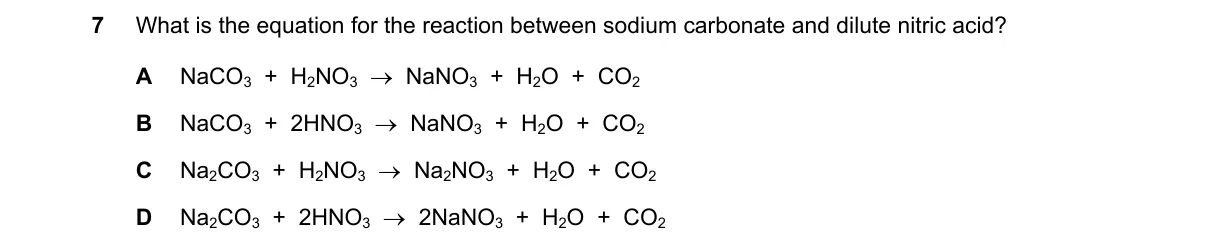
Explanation
Balancing the acid-carbonate neutralization reaction
Steps:
- Write the reactants: sodium carbonate is Na₂CO₃, dilute nitric acid is HNO₃.
- Identify products: salt (NaNO₃), water (H₂O), and carbon dioxide (CO₂).
- Balance sodium: 2 Na on left requires 2 NaNO₃ on right.
- Balance hydrogen and nitrate: 2 H and 2 NO₃ on left require 2 HNO₃; this yields H₂O and CO₂.
Why D is correct:
- It follows the stoichiometry of the double displacement reaction, where one carbonate ion reacts with two nitrate ions and two hydrogens to form two sodium nitrates, water, and CO₂.
Why the others are wrong:
- A: Uses incorrect formula NaCO₃ (sodium carbonate is Na₂CO₃) and is unbalanced.
- B: Produces only one NaNO₃, violating charge and atom balance.
- C: Uses one HNO₃, insufficient for balancing sodium and hydrogen atoms.
Final answer: D
Topic: Preparation of salts
Practice more O Levels Chemistry (5070) questions on mMCQ.me