O Levels Chemistry (5070)•5070/12/M/J/22
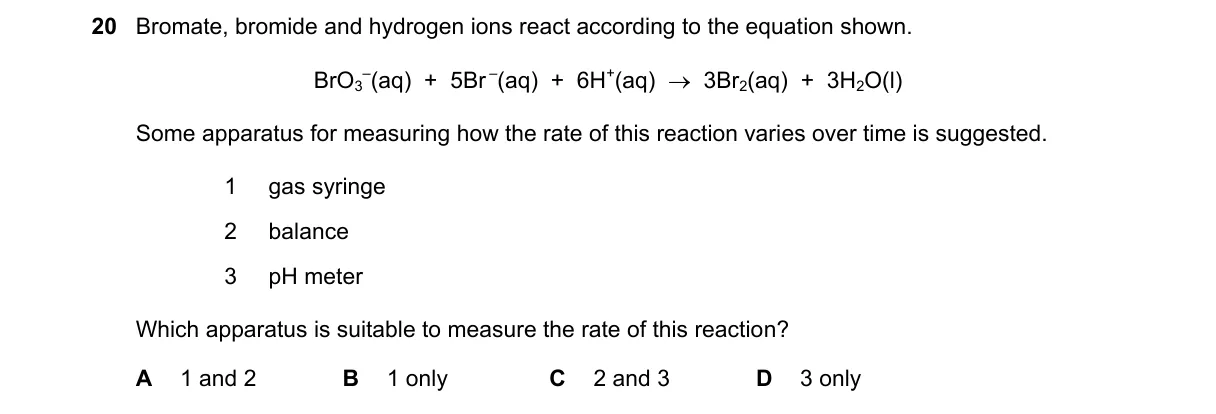
Explanation
Monitoring reaction rate via mass loss from volatile product
Steps:
- Analyze the equation: No gaseous products formed, only Br₂(aq) and H₂O(l).
- Assess gas syringe: Cannot measure volume change without gas production.
- Assess pH meter: H⁺ consumption causes pH rise, but logarithmic scale complicates linear rate calculation.
- Assess balance: Volatile Br₂ evaporates in open system, causing measurable mass loss proportional to reaction progress.
Why D is correct:
- Balance tracks mass loss rate, directly tied to Br₂ production stoichiometry (3 mol Br₂ per reaction), allowing rate determination via Δm/Δt.
Why the others are wrong:
- A: Includes unsuitable gas syringe and imprecise pH monitoring.
- B: Gas syringe useless without gas evolution.
- C: pH meter's nonlinear response hinders accurate rate variation over time.
Final answer: D
Topic: Rate of reaction
Practice more O Levels Chemistry (5070) questions on mMCQ.me