O Levels Chemistry (5070)•5070/12/M/J/20
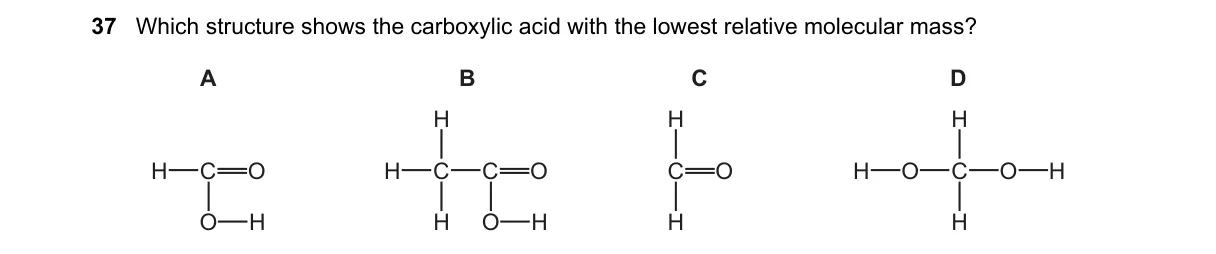
Explanation
Carboxylic acids with lowest molecular mass have shortest carbon chains
Steps:
- Identify the general formula for carboxylic acids: R-COOH, where R is a hydrocarbon chain.
- Calculate relative molecular mass (Mr) for each option using atomic masses (C=12, H=1, O=16).
- Compare Mr values: smallest chain yields lowest Mr.
- Select the structure with the shortest chain (fewest carbons).
Why A is correct:
- A represents formic acid (HCOOH), with Mr = (1×1 + 1×12 + 2×16 + 1×1) = 46, the smallest possible for a carboxylic acid per IUPAC definition.
Why the others are wrong:
- B (acetic acid, CH3COOH) has Mr=60 due to extra carbon, higher than A.
- C (propanoic acid, C2H5COOH) has Mr=74 from longer chain.
- D (butanoic acid, C3H7COOH) has Mr=88, largest chain shown.
Final answer: A
Topic: Carboxylic acids
Practice more O Levels Chemistry (5070) questions on mMCQ.me