O Levels Chemistry (5070)•5070/12/M/J/20
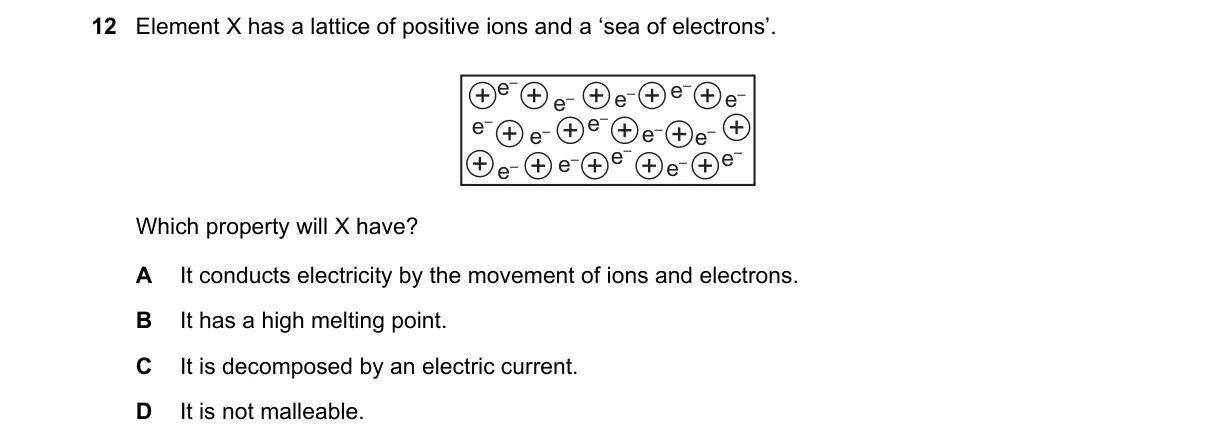
Explanation
Metallic Bonding in Element X
Steps:
- Identify the description "lattice of positive ions and a sea of electrons" as metallic bonding, characteristic of metals.
- Recall that metallic bonds involve strong electrostatic attractions between cations and delocalized electrons.
- List key metal properties: high melting points, electrical conductivity via electrons, malleability, and stability under current.
- Match properties to options, selecting the one directly tied to bond strength.
Why B is correct:
- Metallic bonds form a strong lattice requiring high energy to overcome, leading to high melting points as per the definition of metallic bonding.
Why the others are wrong:
- A: Metals conduct via mobile electrons only; ions remain fixed in the lattice.
- C: Metals are not decomposed by electric current; this applies to electrolytes in electrolysis.
- D: Metals are malleable due to layers of ions sliding past each other.
Final answer: B
Topic: Metallic bonding
Practice more O Levels Chemistry (5070) questions on mMCQ.me