O Levels Chemistry (5070)•5070/11/M/J/20
Explanation
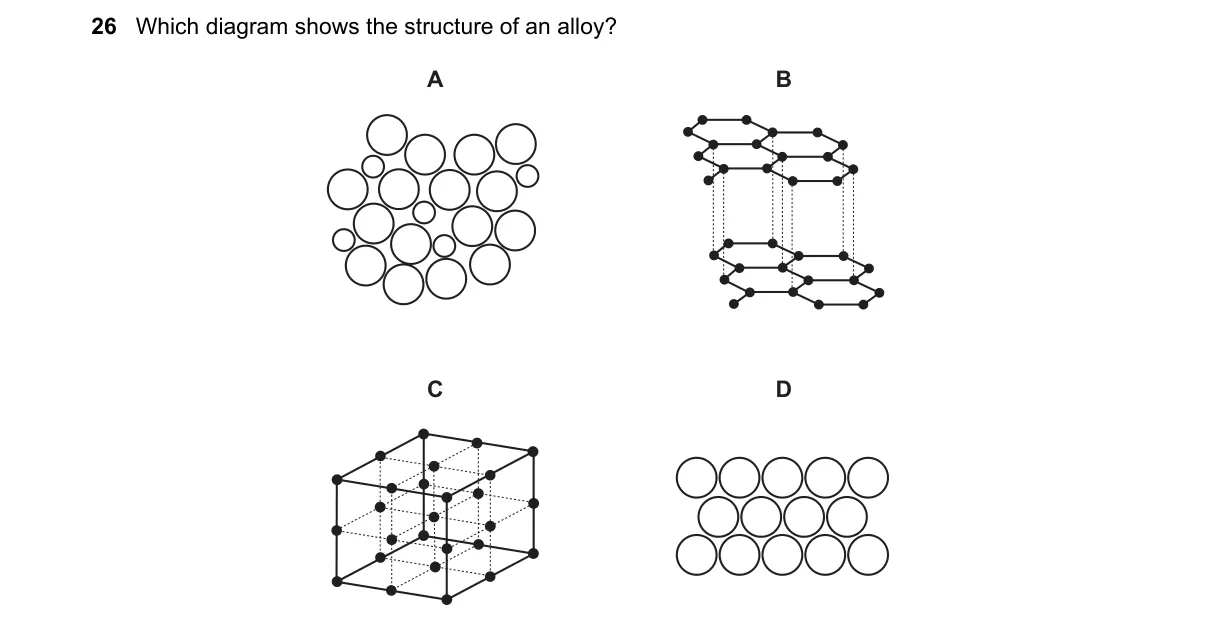
Alloys exhibit irregular atomic arrangements in a metallic lattice
Steps:
- Define alloy as a homogeneous mixture of two or more metals or a metal and non-metal.
- Recall that alloy structures show atoms randomly substituting or interstitially placed in a lattice, unlike ordered pure metals or compounds.
- Examine diagrams: identify irregular mixing in one option versus regular patterns in others.
- Select the diagram depicting random atomic distribution as the alloy structure.
Why A is correct:
- A illustrates a substitutional alloy where different metal atoms randomly occupy lattice sites, matching the definition of a solid solution alloy.
Why the others are wrong:
- B shows a regular, uniform lattice typical of a pure metal crystal.
- C depicts an ordered ionic compound with distinct cation-anion arrangement.
- D represents a molecular structure with discrete units, not metallic bonding.
Final answer: A
Topic: Alloys and their properties
Practice more O Levels Chemistry (5070) questions on mMCQ.me