O Levels Chemistry (5070)•5070/11/M/J/20
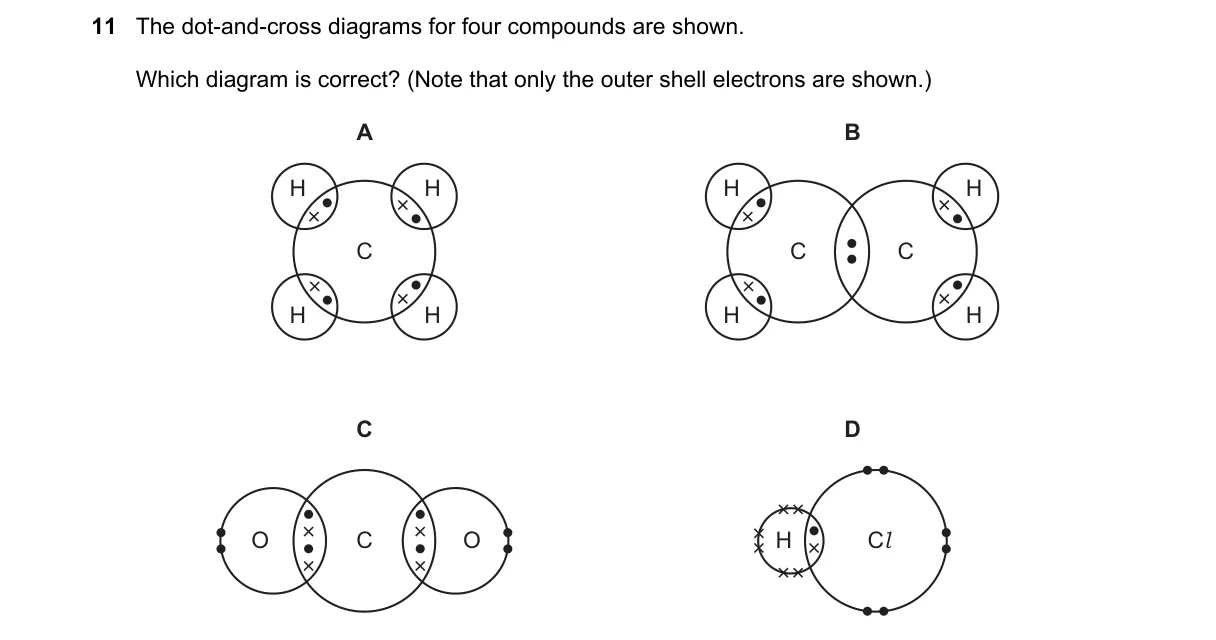
Explanation
Lewis Dot-and-Cross Diagrams Follow Octet Rule
Steps:
- Count total valence electrons for the compound using atomic numbers.
- Sketch skeletal structure with central atom bonded to others.
- Distribute electrons as lone pairs and shared pairs to satisfy octets.
- Verify all atoms achieve stable electron configurations.
Why A is correct:
- Diagram A shows correct shared and lone pairs, adhering to the octet rule where each atom has 8 valence electrons (except hydrogen with 2).
Why the others are wrong:
- B incorrectly shows unpaired electrons on central atom, violating octet rule.
- C has excessive lone pairs, exceeding total valence electrons available.
- D misplaces bonds, resulting in incomplete octet for oxygen.
Final answer: A
Topic: Simple molecules and covalent bonds
Practice more O Levels Chemistry (5070) questions on mMCQ.me