O Levels Chemistry (5070)•5070/12/M/J/19
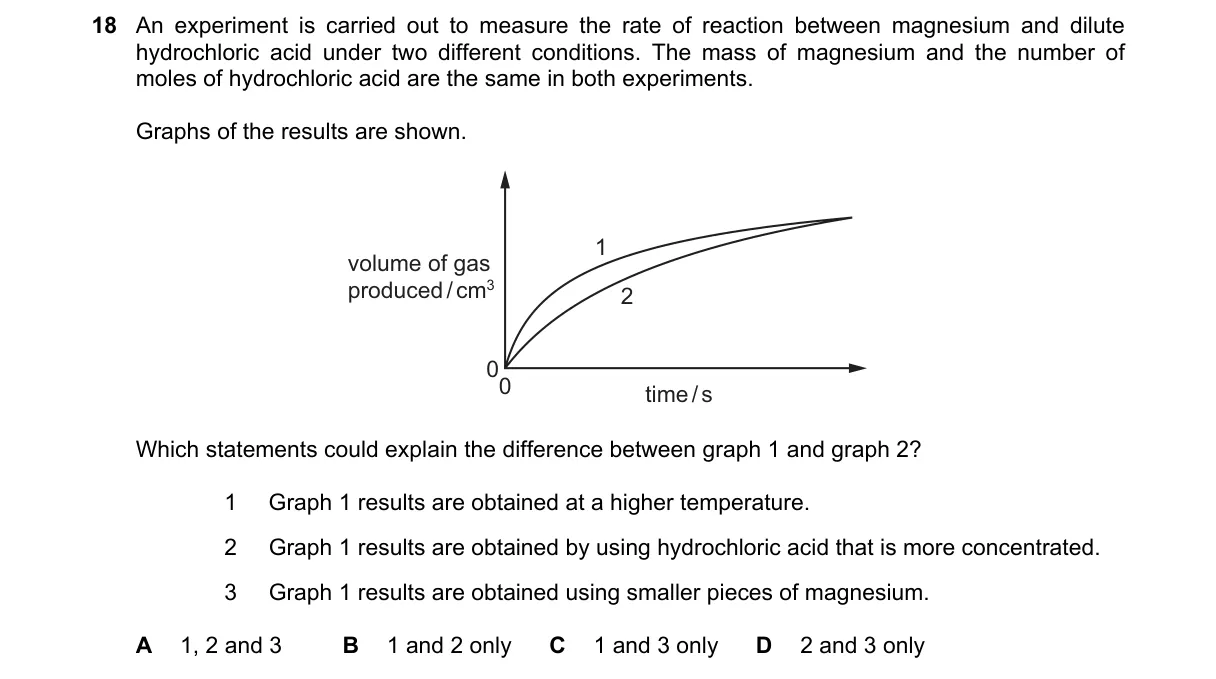
Explanation
Factors Influencing Reaction Rate in Mg + HCl
Steps:
- Reaction rate increases with HCl concentration due to more frequent collisions.
- Smaller magnesium pieces increase surface area, raising collision frequency and rate for graph 1.
- Fewer magnesium pieces (same mass) mean larger pieces, reducing surface area and slowing rate for graph 1.
- All three factors alter either concentration or surface area, explaining differences between graphs.
Why A is correct:
- Collision theory states reaction rate depends on reactant concentration and effective collisions, which surface area affects; all statements modify these.
Why the others are wrong:
- B omits statement 3, which reduces surface area and can slow the reaction.
- C omits statement 2, which increases surface area and can speed up the reaction.
- D omits statement 1, ignoring concentration's direct impact on collision rate.
Final answer: A
Topic: Rate of reaction
Practice more O Levels Chemistry (5070) questions on mMCQ.me