O Levels Chemistry (5070)•5070/11/M/J/19
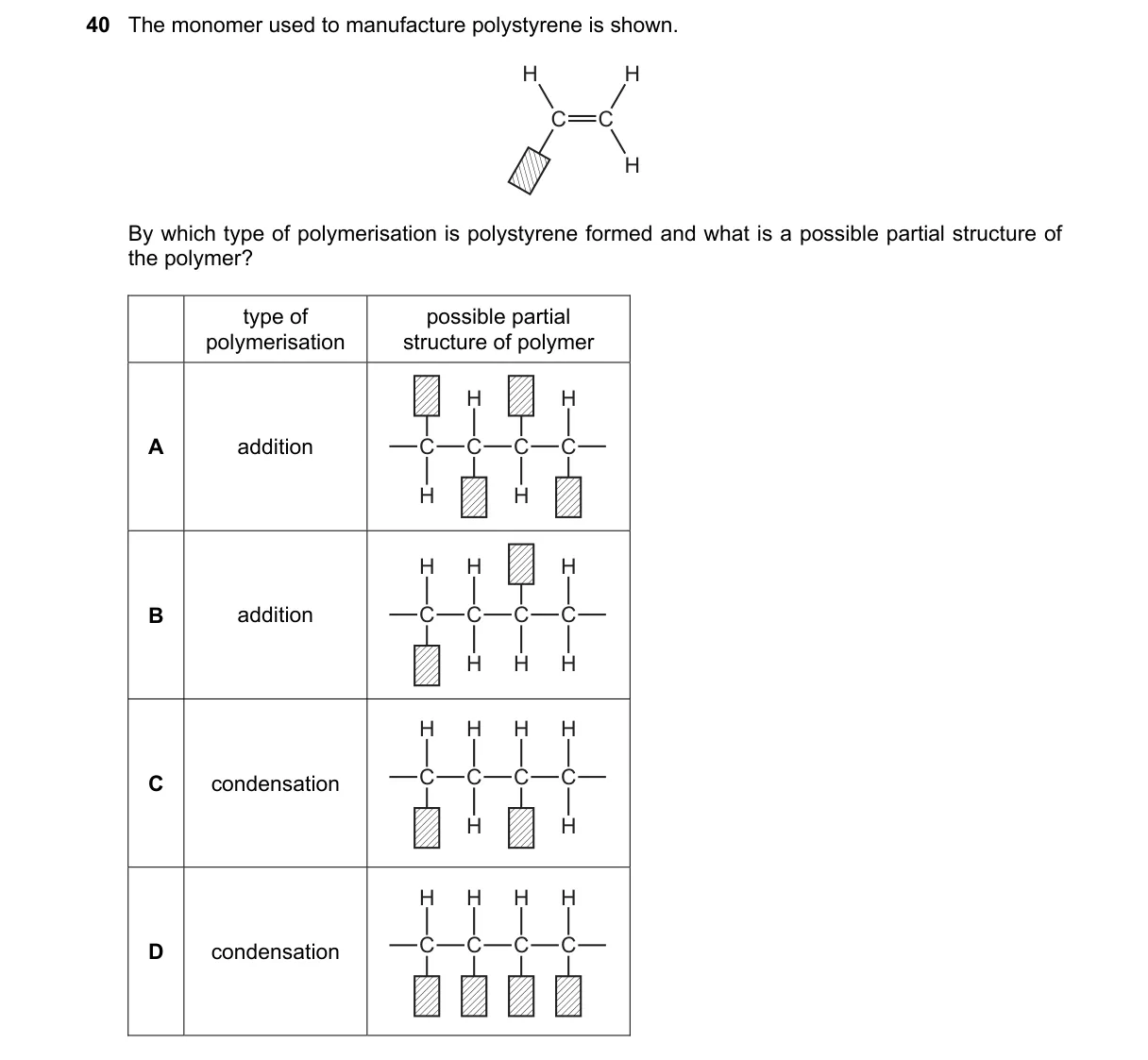
Explanation
Polystyrene via Addition Polymerization Steps:
- Identify monomer as styrene (C6H5-CH=CH2) with a reactive vinyl group.
- Recognize addition polymerization opens the C=C double bond to form chains without byproducts.
- Confirm polystyrene's structure: repeating [-CH2-CH(C6H5)-] units from styrene linkage.
- Rule out condensation, which requires functional groups like -OH or -COOH for small molecule elimination.
Why B is correct:
- B pairs addition polymerization with the accurate partial structure of polystyrene, matching the vinyl addition mechanism.
Why the others are wrong:
- A: Addition type correct, but likely shows incorrect partial structure (e.g., wrong linkage).
- C: Condensation incorrect; styrene lacks groups for elimination reactions like water loss.
- D: Condensation incorrect and partial structure mismatched for polystyrene.
Final answer: B
Topic: Polymers
Practice more O Levels Chemistry (5070) questions on mMCQ.me