O Levels Chemistry (5070)•5070/11/M/J/19
Explanation
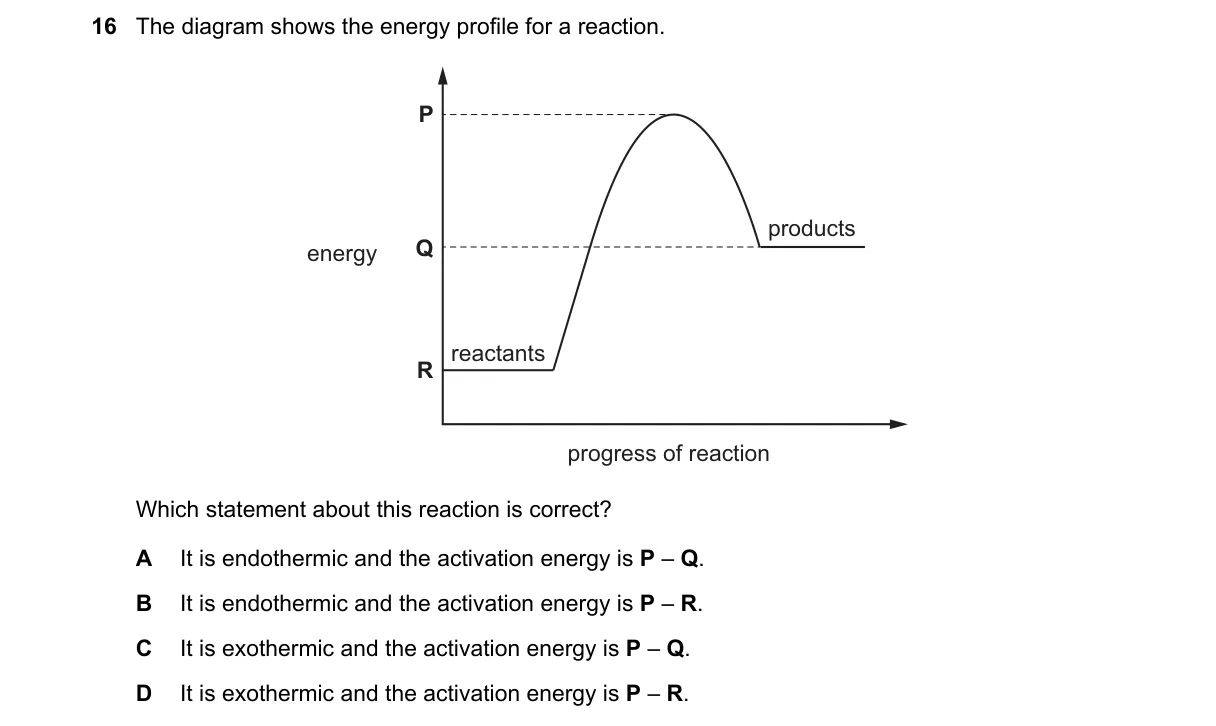
Interpreting Reaction Energy Profiles
Steps:
- Compare reactant and product energy levels: if products are lower, the reaction is exothermic.
- Locate the transition state at the profile's peak energy.
- Activation energy (Ea) is the energy difference from reactants to the transition state peak.
- Match labels: P as reactant energy, R as peak (transition state), Q as product energy (lower than P).
Why B is correct:
- Exothermic reactions release energy (products lower than reactants, per ΔH < 0 definition); Ea = P-R matches the barrier from reactants (P) to peak (R).
Why the others are wrong:
- A: Incorrectly labels as endothermic (products not higher).
- C: Wrong type (endothermic) and Ea (Q-R misidentifies barrier).
- D: Correct type but wrong Ea (R-P reverses direction, yielding negative value).
Final answer: B
Topic: Exothermic and endothermic reactions
Practice more O Levels Chemistry (5070) questions on mMCQ.me