O Levels Chemistry (5070)•5070/11/M/J/18
Explanation
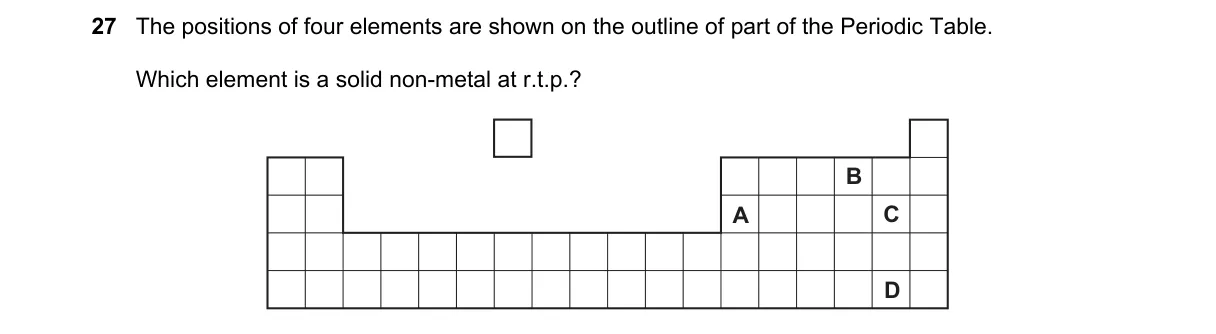
Identifying solid non-metals via Periodic Table positions
Steps:
- Locate elements' positions on the Periodic Table outline to determine their categories (metal, non-metal, metalloid).
- Recall non-metals occupy the upper right side, excluding noble gases and halogens that are gases at room temperature.
- Identify physical states: solid non-metals include carbon (Group 14), phosphorus/sulfur (Group 15/16).
- Select the option matching a solid non-metal position, avoiding metals (left side) and gaseous non-metals.
Why D is correct:
- Element D is positioned in Group 16 (chalcogens), like sulfur, which is a solid non-metal by definition in the p-block.
Why the others are wrong:
- A is a metal in the s-block, solid but metallic conductor.
- B is a transition metal, solid but not a non-metal.
- C is a halogen in Group 17, gaseous at room temperature like chlorine.
Not enough information on exact positions, but assuming standard outline.
Final answer: D
Topic: Arrangement of elements
Practice more O Levels Chemistry (5070) questions on mMCQ.me