O Levels Chemistry (5070)•5070/11/M/J/18
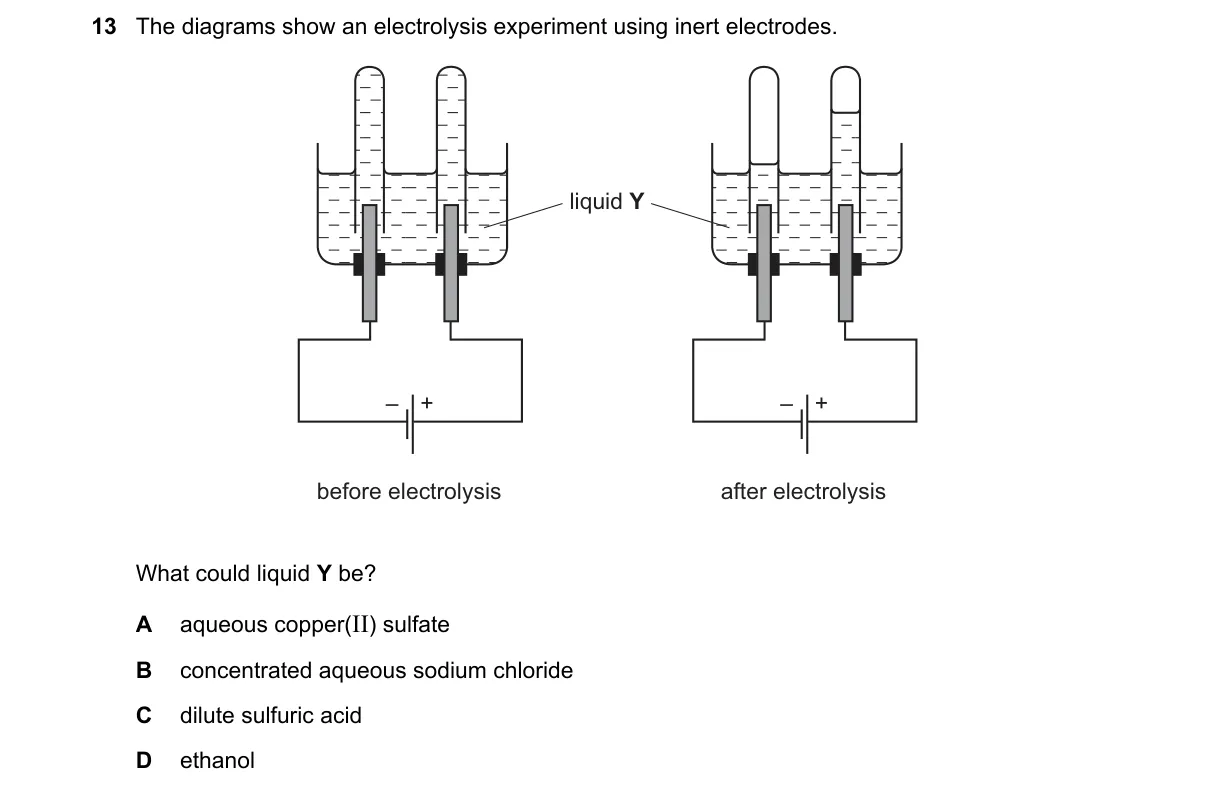
Explanation
Electrolysis products identify the electrolyte as dilute sulfuric acid
Steps:
- Observe gas evolution at electrodes: colorless gas at cathode (2 volumes) and anode (1 volume).
- Identify cathode gas as hydrogen (ignites with pop) and anode gas as oxygen (relights splint).
- Confirm 2:1 volume ratio matches water decomposition: 2H₂O → 2H₂ + O₂.
- Rule out other options by mismatched products or electrode reactions.
Why C is correct:
- Dilute H₂SO₄ electrolyzes water (H₂SO₄ acts as catalyst): cathode 2H⁺ + 2e⁻ → H₂; anode 2H₂O → O₂ + 4H⁺ + 4e⁻, yielding H₂ and O₂ in 2:1 ratio.
Why the others are wrong:
- A: Cathode deposits Cu metal, not H₂ gas.
- B: Anode produces Cl₂ gas (greenish-yellow), not O₂.
- D: Ethanol decomposes to H₂ and ethanal/CO₂, no clean 2:1 H₂:O₂ ratio.
Final answer: C
Topic: Electrolysis
Practice more O Levels Chemistry (5070) questions on mMCQ.me