O Levels Physics (5054)•5054/12/O/N/21
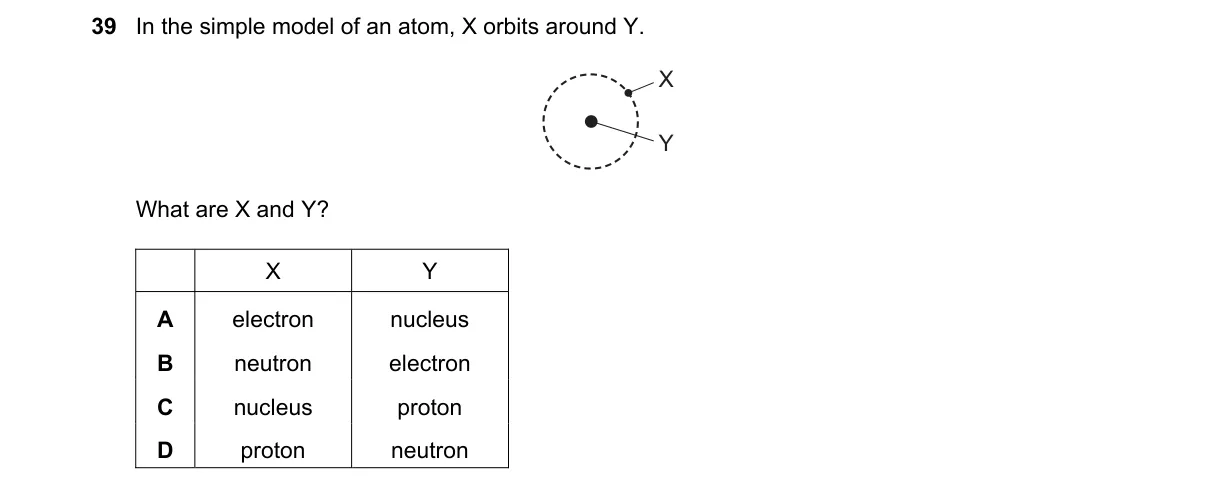
Explanation
Bohr Model of the Atom Steps:
- Recall the simple atomic model where particles orbit a central core.
- Identify the central core as the nucleus, containing protons and neutrons.
- Recognize electrons as negatively charged particles orbiting the nucleus.
- Match the phrase "X is around Y" to electrons surrounding the nucleus.
Why A is correct:
- In the Bohr model, electrons orbit the nucleus, forming electron shells around the positively charged core.
Why the others are wrong:
- B: Neutrons reside in the nucleus, not around electrons.
- C: The nucleus contains protons but is not around them.
- D: Protons and neutrons both form the nucleus, not one around the other.
Final answer: A
Topic: The atom
Practice more O Levels Physics (5054) questions on mMCQ.me